To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Total air temperatureTotal air temperature is a term used generally in aviation. It is measured by a specially designed temperature probe mounted on the surface of the aircraft. The probe is designed to bring the air to rest relative to the aircraft. As the air is brought to rest kinetic energy is converted to internal energy. The air is compressed and experiences an adiabatic increase in temperature. Therefore total air temperature is higher than the static (or ambient) air temperature. Product highlightTotal air temperature is an essential input to an air data computer in order to enable computation of static air temperature and hence true airspeed. The relationship between static and total air temperatures is given by:
where: T0 = static air temperature (kelvin or degree Rankine)
Where: e = recovery factor (also noted Ct)
Platinum wire ratiometer thermometer ("flush bulb type"): e ≈ 0.75 - 0.9 Double platinum tube ratiometer thermometer ("TAT probe"): e ≈ 1 Other notations Total air temperature (TAT) is also called: indicated air temperature (IAT) or ram air temperature (RAT) The difference between TAT & SAT is called ram rise (RR) In practice the ram rise is negligible for aircraft flying at (true) airspeeds under Mach 0.2 |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Total_air_temperature". A list of authors is available in Wikipedia. |





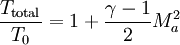
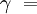 ratio of specific heats = approx 1.4 for air
ratio of specific heats = approx 1.4 for air