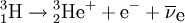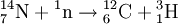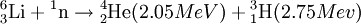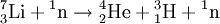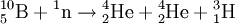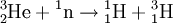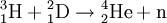To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Tritium
Tritium (symbol T or ³H) is a radioactive isotope of hydrogen. The nucleus of tritium (sometimes called a triton) contains one proton and two neutrons, whereas the nucleus of protium (the most abundant hydrogen isotope) contains no neutrons. Product highlight
DecayTritium is radioactive with a half-life of 12.32 years. It decays into helium-3 by the reaction releasing 18.6 keV of energy. The electron has an average kinetic energy of 5.7 keV, while the remaining energy is carried off by the nearly undetectable electron antineutrino. The low-energy beta radiation from tritium cannot penetrate human skin, so tritium is only dangerous if inhaled or ingested. Its low energy also creates difficulty detecting tritium labelled compounds except by using liquid scintillation counting. ProductionTritium occurs naturally due to cosmic rays interacting with atmospheric gases. In the most important reaction for natural tritium production, a fast neutron (> 4MeV [1]) interacts with atmospheric nitrogen: Because of tritium's relatively short half-life, however, tritium produced in this manner does not accumulate over geological timescales, and its natural abundance is negligible. Tritium is produced in nuclear reactors by neutron activation of lithium-6. This is possible with neutrons of any energy, and is an exothermic reaction yielding 4.8 MeV, which is more than one-quarter of the energy that fusion of the produced triton with a deuteron can later produce. High-energy neutrons can also produce tritium from lithium-7 in an endothermic reaction, consuming 2.822 MeV. This was discovered when the 1954 Castle Bravo nuclear test produced an unexpectedly high yield.[2] High-energy neutrons irradiating boron-10 will also occasionally produce tritium. [3]The more common result of boron-10 neutron capture is 7Li and a single alpha particle.[4] The reactions requiring high neutron energies are not attractive production methods. Tritium's decay product helium-3 has a very large cross section for the (n,p) reaction with thermal neutrons and is rapidly converted back to tritium in a nuclear reactor. Tritium is occasionally a direct product of nuclear fission, with a yield of about 0.01% (one per 10000 fissions).[5][6] This means that tritium release or recovery needs to be considered in nuclear reprocessing even in ordinary spent nuclear fuel where tritium production was not a goal. Tritium is also produced in heavy water-moderated reactors when deuterium captures a neutron. This reaction has a very small cross section (which is why heavy water is such a good neutron moderator) and relatively little tritium is produced; nevertheless, cleaning tritium from the moderator may be desirable after several years to reduce the risk of escape to the environment. According to IEER's 1996 report about the United States Department of Energy, only 225 kg of tritium has been produced in the US since 1955. Since it is continuously decaying into helium-3, the stockpile was estimated as approximately 75 kg at the time of the report.[7] Tritium for American nuclear weapons was produced in special heavy water reactors at the Savannah River Site until their shutdown in 1988; with the Strategic Arms Reduction Treaty after the end of the Cold War, existing supplies were sufficient for the new, smaller number of nuclear weapons for some time. Production was resumed with irradiation of lithium-containing rods (replacing the usual boron-containing control rods) at the commercial Watts Bar Nuclear Generating Station in 2003-2005 followed by extraction of tritium from the rods at the new Tritium Extraction Facility at SRS starting in November 2006.[8] PropertiesTritium has an atomic mass of 3.0160492. It is a gas (T2 or ³H2) at standard temperature and pressure. Tritium combines with oxygen to form a liquid called tritiated water T2O or partially tritiated THO. Tritium figures prominently in studies of nuclear fusion due to its favorable reaction cross section and the high energy yield of 17.6 MeV for its reaction with deuterium: All atomic nuclei, being composed of protons and neutrons, repel one another because of their positive charge. However, if the atoms have a high enough temperature and pressure (as is the case in the core of the Sun, for example), then their random motions can overcome such electrical repulsion (called the Coulomb force), and they can come close enough for the strong nuclear force to take effect, fusing them into heavier atoms. Since tritium has the same charge as ordinary hydrogen, it experiences the same electrostatic repulsive force (see Coulomb's law). However, due to tritium's supply of neutrons which are carried into reactions and feel the attractive strong force once delivered, tritium can more easily fuse with other light atoms. The same is also true, albeit to a lesser extent, of deuterium, and that is why brown dwarfs (so-called failed stars) cannot burn hydrogen, but do indeed burn deuterium. Before the onset of atmospheric nuclear weapons tests, the global equilibrium tritium inventory was estimated at about 80 megacuries (MCi). Like hydrogen, tritium is difficult to confine; rubber, plastic, and some kinds of steel are all somewhat permeable. This has raised concerns that if tritium is used in quantity, in particular for fusion reactors, it may contribute to radioactive contamination, although its short half-life should prevent any significant accumulation in the atmosphere. Atmospheric nuclear testing (prior to the Partial Test Ban Treaty) proved unexpectedly useful to oceanographers, as the sharp spike in surface tritium levels could be used over the years to measure the rate at which the lower and upper ocean levels mixed. Regulatory limitsThe legal limits for tritium in drinking water can vary. The U.S. limit is calculated to yield a dose of 4 mrem (or 40 microsieverts in SI units) per year.
UsageSelf-powered lightingThe emitted electrons from small amounts of tritium cause phosphors to glow so as to make self-powered lighting devices called trasers which are now used in watches and exit signs. It is also used in certain countries to make glowing keychains, and compasses. In recent years, the same process has been used to make self-illuminating gun sights for firearms. These take the place of radium, which can cause bone cancer. These uses of radium have been banned in most countries for decades. The aforementioned IEER report claims that the commercial demand for tritium is 400 grams per year. Nuclear weaponsTritium is widely used in nuclear weapons for boosting a fission bomb or the fission primary of a thermonuclear weapon. Before detonation, a few grams of tritium-deuterium gas are injected into the hollow "pit" of fissile plutonium or uranium. The early stages of the fission chain reaction supply enough heat and compression to start DT fusion, then both fission and fusion proceed in parallel, the fission assisting the fusion by continuing heating and compression, and the fusion assisting the fission with highly energetic (14.1 MeV) neutrons. As the fission fuel depletes and also explodes outward, it falls below the density needed to stay critical by itself, but the fusion neutrons make the fission process progress faster and continue longer than it would without boosting. Increased yield comes overwhelmingly from the increase in fission; the energy released by the fusion itself is much smaller because the amount of fusion fuel is much smaller. Besides increased yield (for the same amount of fission fuel with vs. without boosting) and the possibility of variable yield (by varying the amount of fusion fuel), possibly even more important advantages are allowing the weapon (or primary of a weapon) to have a smaller amount of fissile material (eliminating the risk of predetonation by nearby nuclear explosions) and more relaxed requirements for implosion, allowing a smaller implosion system. Because the tritium in the warhead is continuously decaying, it is necessary to replenish it periodically. The estimated quantity needed is 4 grams per warhead.[9] To maintain constant inventory, 0.22 grams per warhead per year must be produced. As tritium quickly decays and is difficult to contain, the much larger secondary charge of a thermonuclear weapon instead uses lithium deuteride as its fusion fuel; during detonation, neutrons split lithium-6 into helium-4 and tritium; the tritium then fuses with deuterium, producing more neutrons. As this process requires a higher temperature for ignition, and produces fewer and less energetic neutrons (only D-D fusion and 7Li splitting are net neutron producers), LiD is not used for boosting, only for secondaries.
Controlled nuclear fusionTritium is an important fuel for controlled nuclear fusion in both magnetic confinement and inertial confinement fusion reactor designs. The experimental fusion reactor ITER and the National Ignition Facility (NIF) will use Deuterium-Tritium (D-T) fuel. The D-T reaction is favored since it has the largest fusion cross-section (~ 5 barns peak) and reaches this maximum cross-section at the lowest energy (~65 keV center-of-mass) of any potential fusion fuel. History
Tritium was first predicted in the late 1920s by Walter Russell, using his "spiral" periodic table[citation needed], then produced in 1934 from deuterium, another isotope of hydrogen, by Ernest Rutherford, working with Mark Oliphant and Paul Harteck. Rutherford was unable to isolate the tritium, a job that was left to Luis Alvarez and Robert Cornog, who correctly deduced that the substance was radioactive. Willard F. Libby discovered that tritium could be used for dating water, and therefore wine. Tritium was seen in Spider-man 2 when "Doc Ock" used it to (unsuccessfully) sustain a nuclear fusion reaction. References
Categories: Isotopes of hydrogen | Environmental isotopes | Radiochemistry | Radioisotope fuels | Nuclear fusion fuels |
|||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Tritium". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||||||||||||||