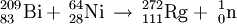To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Roentgenium
Roentgenium (pronounced /rɛntˈgɛniəm/, /rəntˈjɛniəm/, formerly Unununium /ˌjuːnəˈnʌniəm/) is a chemical element in the periodic table that has the symbol Rg and atomic number 111, making it one of the super-heavy atoms. It is a synthetic element whose longest-lived isotope has a mass of 280 and a half-life of 3.6 seconds. Due to its presence in Group 11, it is a transition metal. Additional recommended knowledgeHistoryIt was discovered by Peter Armbruster, Gottfried Münzenber, and their team working at the Gesellschaft für Schwerionenforschung (GSI) in Darmstadt, Germany on December 8, 1994. Only three atoms of it were observed (all 272Rg), by the fusion of 209Bismuth and 64Nickel in a linear accelerator (Nickel was bombarded onto the bismuth target): The name roentgenium was accepted as a permanent name on November 1 2004 in honor of Wilhelm Conrad Röntgen; before this date, the element was known under the temporary IUPAC systematic element name unununium, symbol Uuu). Some research has referred to it as eka-gold. IsotopesTwelve isotopes of roentgenium are known. The longest-lived of these is 280Rg, which decays through alpha decay and has a half-life of 3.6 seconds. The shortest-lived isotope is 272Rg, which decays through alpha decay and has a half life of 1.5 ms. Another known isotope, 279Rg, decays through alpha decay and has a half-life of 170 ms. See alsoCategories: Chemical elements | Transition metals |
|||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Roentgenium". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||||||||||||||||||