To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Arsenic trisulfide
Arsenic trisulfide is the chemical compound with the formula As2S3. This bright yellow solid is well known because it occurs as the mineral orpiment, has been used as a pigment, and has played a role in the analysis of arsenic compounds. This chalcogenide material is a group V/VI, intrinsic p-type, semiconductor and exhibits photo-induced phase-change properties. The other important As-S phase is realgar, As4S4, which is red-orange and also occurs as a mineral. Product highlight
StructureAs2S3 occurs both in crystalline and amorphous forms. Both forms feature polymeric structures consisting As(III) centers are three-fold co-ordinated by sulfide centers in a trigonal pyramidal geometery. The sulfide centres are two-fold coordinated to 2 arsenic atoms. In the crystalline form, the As-S bonds form bonded sheets.[1] The bonding between the sheets consists of van der Waals forces. The crystalline form is usually found in geological samples. Amorphous As2S3 does not possess s layered structure but is more highly cross-linked. Like other glasses, there is no medium or long-range order, but the first co-ordination sphere is well defined. As2S3 is a good glass former and exhibits a wide glass-forming region in its phase diagram. Synthesis and reactionsFrom the elementsAmorphous As2S3 is obtained via the fusion of the elements at 390 °C. Rapid cooling of the reaction melt ensures the disordered arrangement of the bonds, resulting in the glass. Like other chemical syntheses, pure precursors and the exclusion of air provides purer product. The reaction can be represented with the chemical equation:
Upon heating in a vacuum, polymeric As2S3 "cracks" to give a mixture of molecular species, including molecular As4S6.[2][3] As4S6 adopts the adamantane geometry, like that observed for P4O6 and As4O6. When a film of this material is exposed to an external energy source such as thermal energy (via thermal annealing [4]), electromagnetic radiation (i.e. UV lamps, lasers[5], electron beams)[6]), As4S6 polymerizes:
Aqueous precipitation
As2S3 forms when aqueous solutions containing As(III) are treated with H2S. Arsenic was in the past analyzed and assayed by this reaction, which results in the precipitation of As2S3, which is then weighed. As2S3 can even be precipitated in 6M HCl. As2S3 is so insoluble that it is not toxic. As2S3 characteristically dissolves upon treatment with aqueous solutions containing sulfide ions. The dissolved arsenic species is the pyramidal trianion AsS33-:
As2S3 is the anhydride of the hypothetical thioarsenous acid, As(SH)3. Upon treatment with polysulfide ions, As2S3 dissolves to give a variety of species containing both S-S and As-S bonds. One of the more noteworthy derivatives is S7As-S-, a ring that contains an exocyclic sulfido center attached to the As atom. As2S3 also dissolves in strongly alkaline solutions to give a mixture of AsS33- and AsO33-.[7] Reactions with oxygen"Roasting" As2S3 in air gives volatile, toxic derivatives, this conversion being one of the hazards associated with the refining of heavy metal ores:
Contemporary usesAs an inorganic photoresistDue to its high refractive index of 2.45 and its large Knoop hardness compared to organic photoresists, As2S3 has been investigated for the fabrication of photonic crystals with a full-photonic band-gap. Advances in laser patterning techniques such as three-dimensional direct laser writing (3-D DLW) and chemical wet-etching chemistry, has allowed this material to be used as a photoresist to fabricate 3-D nanostructures.[8][9] As2S3 has been investigated for use as a high resolution photoreist material since the early 1970's,[10][11] using aqueous etchants. Although these aqueous etchants allowed for low-aspect ratio 2-D structures to be fabricated, they do not allow for the etching of high aspect ratio structures with 3-D periodicity. Certain organic reagents, used in organic solvents, permit the high-etch selectivity required to produce high-aspect ratio structures with 3-D periodicity. Medical applicationsAs2S3 and As4S4 have been investigated as treatments for acute promyelocytic leukemia (APL).[12] The mode of action is thought to be similar to that for As2O3. For IR-transmitting glassesArsenic trisulfide manufactured into amorphous form is used as a chalcogenide glass for infrared optics. It is transparent between 620 nm and 11 µm. The arsenic trisulfide glass is more resistant to oxidation than crystalline arsenic trisulfide, which minimizes toxicity concerns.[1] It can be also used as an acousto-optic material. Role in ancient artistryThe ancient Egyptians reportedly used orpiment, natural or synthetic, as a pigment in artistry and cosmetics. MiscellaneousArsenic trisulfide is also used as a tanning agent. It was formerly used with indigo dye for the production of pencil blue, which allowed dark blue hues to be added to fabric via pencil or brush. Precipitation of arsenic trisulfide is used as an analytical test for presence of dissimilatory arsenic-reducing bacteria (DARB).[13] SafetyAs2S3 is so insoluble that it is considered non-toxic. Crystalline As2S3 however tends to oxidize on the surface, forming a layer of toxic arsenic trioxide. Commercial orpiment can however contain substantial amounts of arsenic oxides, which are soluble and therefore highly toxic. References
Categories: Sulfides | Arsenic compounds |
|||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Arsenic_trisulfide". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||||||||||||||||||





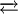 As4S6
As4S6


