To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Capillary lengthIn fluid mechanics, capillary length is a characteristic length scale for fluid subject to a body force from gravity and a surface force due to surface tension. Product highlightThe capillary length is defined as[1]:
where g is the acceleration due to gravity and ρ is the density of the fluid, and γ is the surface tension of the fluid-fluid interface. For clean water at standard temperature and pressure, the capillary length is ~2mm. A capillary surface that has a characteristic length smaller than the capillary length can be considered a low Bond number surface. A sessile drop whose largest dimension is smaller than the capillary length, for example, will take the shape of spherical cap, which is the solution to the Young-Laplace equation with gravity completely absent. See alsoReferences
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Capillary_length". A list of authors is available in Wikipedia. |





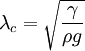 ,
,


