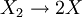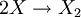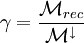To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
CatalycityCatalycity is a macroscopic parameter to describe surface catalysis (heterogeneous catalysis). It is usually called a material property, due to its strong dependence on the surface material. This naming is not fully justified, since the catalycity not only dependent on the material but also on its environment.
Product highlight
CatalysisGases like Nitrogen, Oxygen or mixtures of the like Air consists at low temperatures of molecules. At elevated temperatures they dissociate, i.e. they break apart into single atoms.
This process can be accelerated with the help of a catalyst. The catalyst provides an alternative reaction path, which is in general faster than the original.
Such and reaction path could be: 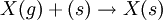 reaction at the surface: 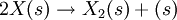 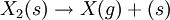
DefinitionCatalyctiy relates the number of reacting atoms to the number of atoms that hit a surface. Therefore, a catalycity of one means that all impinging particles do recombine.
UsageThe parameter catalycity is mainly used in hypersonics, i.e. the science high speed and high temperature flow. These kind of flows occur for instance in Atmospheric reentry. LimitationsIn mixtures of gases where interactions between the different species occurs, the parameter catalycity is not appropriate. DeterminationThe determination of catalycity is usually done in Plasma (physics) wind tunnels like arc-jets or inductively coupled plasma generators. Categories: Chemical kinetics | Surface chemistry | Chemical engineering |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Catalycity". A list of authors is available in Wikipedia. |