To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Chemical affinityIn physical chemistry, chemical affinity can be defined as electronic properties by which dissimilar chemical species are capable of forming chemical compounds.[1] Chemical affinity can also refer to the tendency of an atom or compound to combine by chemical reaction with atoms or compounds of unlike composition. According to chemistry historian Henry Leicester, the influential 1923 textbook Thermodynamics and the Free Energy of Chemical Reactions by Gilbert N. Lewis and Merle Randall led to the replacement of the term “affinity” by the term “free energy” in much of the English-speaking world. Product highlight
Modern conceptionsIn modern terms, we relate affinity to the phenomenon whereby certain atoms or molecules have the tendency to aggregate or bond. For example, in the 1919 book Chemistry of Human Life physician George W. Carey states: “Health depends on a proper amount of iron phosphate Fe3(PO4)2 in the blood, for the molecules of this salt have chemical affinity for oxygen and carry it to all parts of the organism.” In this antiquated context, chemical affinity is sometimes found synonymous with the term "magnetic attraction". Many writings, up until about 1925, also refer to a “law of chemical affinity”. ThermodynamicsIn 1923, the Belgian mathematician and physicist Théophile de Donder derived a relation between affinity With the writings of Théophile de Donder as precedent, Prigogine and Defay in Chemical Thermodynamics (1954) defined chemical affinity (denoted by
This definition is useful for quantifying the factors responsible both for the state of equilibrium systems (where History"Chemical affinity", historically, refers to the "force" that causes chemical reactions.[2] A broad definition, used generally throughout history, is that chemical affinity is that whereby substances enter into or resist decomposition.[3] In current use, it The following statement, made by Ilya Prigogine, summarizes the concept of affinity:
The term affinity has been used figuratively since c.1600 in discussions of structural relationships in chemistry, philology, etc., and reference to "natural attraction" is from 1616. The idea of affinity is extremely old. Many attempts have been made at identifying its origins.[3] The majority of such attempts, however, except in a general manner, end in futility since ‘affinities’ lie at the basis of all magic, thereby pre-dating science.[4] Physical chemistry, however, was one of the first branches of science to study and formulate a "theory of affinity". The name affinitas was first used in the sense of chemical relation by German philosopher Albertus Magnus near the year 1250. Later, those as Robert Boyle, John Mayow, Johann Glauber, Isaac Newton, and Georg Stahl put forward ideas on elective affinity in attempts to explain how heat is evolved during combustion reactions.[5] The modern term chemical affinity is a somewhat modified variation of its eighteenth-century precursor "elective affinity" or elective attractions, a coinage of the Swedish chemist Torbern Olof Bergman from his book De attractionibus electivis (1775). Antoine Lavoisier, in his famed 1790 Elements of Chemistry, refers to Bergmann’s work and discusses the concept of elective affinities or attractions. Goethe used the concept in his novel Elective Affinities, (1809) Geoffroy's 1718 affinity tableThe first-ever affinity table, which was based on displacement reactions, was published in 1718 by the French chemist Étienne François Geoffroy. Geoffroy's name is best known in connection with these tables of "affinities" (tables des rapports), which were first presented to the French Academy in 1718 and 1720, as shown below: These were lists, prepared by collating observations on the actions of substances one upon another, showing the varying degrees of affinity exhibited by analogous bodies for different reagents, and they retained their vogue for the rest of the century, until displaced by the profounder conceptions introduced by Claude Berthollet. See also
References
Further readingImportant books on Newton's alchemy, as he was one of the main proponents of the theory of chemical affinity, are:
Notes
|
||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Chemical_affinity". A list of authors is available in Wikipedia. |





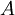 and the
and the 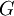 of a chemical reaction. Through a series of derivations, de Donder showed that if we consider a mixture of chemical species with the possibility of chemical reaction, it can be proved that the following relation holds:
of a chemical reaction. Through a series of derivations, de Donder showed that if we consider a mixture of chemical species with the possibility of chemical reaction, it can be proved that the following relation holds:
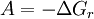
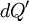 and
and 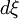 , respectively):
, respectively):
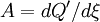 . (1).
. (1).
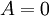 ), and for changes of state of non-equilibrium systems (where
), and for changes of state of non-equilibrium systems (where 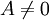 ).
).


