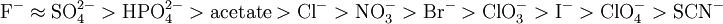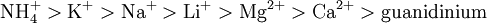To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Hofmeister seriesThe hofmeister series is a classification of ions in order of their ability to change water structure. The effects of these changes were first worked out by Franz Hofmeister, who studied the effects of cations and anions on the solubility of proteins.[1] Product highlightHofmeister discovered a series of salts that have consistent effects on the solubility of proteins and (it was discovered later) on the stability of their secondary and tertiary structure. Anions appear to have a larger effect than cations, and are usually ordered (This is a partial listing; many more salts have been studied.) The order of cations is usually given as The mechanism of the Hofmeister series is not entirely clear, but does not seem to result from changes in general water structure, instead more specific interactions between ions and proteins and ions and the water molecules directly contacting the proteins may be more important.[2] Early members of the series increase solvent surface tension and decrease the solubility of nonpolar molecules ("salt out"); in effect, they strengthen the hydrophobic interaction. By contrast, later salts in the series increase the solubility of nonpolar molecules ("salt in") and increase the order in water; in effect, they weaken the hydrophobic effect. However, these salts also interact directly with proteins (which are charged and have strong dipole moments) and may even bind specifically (e.g., phosphate and sulfate binding to ribonuclease A). Ions that have a strong 'salting in' effect such as I- and SCN- are strong denaturants, because they salt in the peptide group, and thus interact much more strongly with the unfolded form of a protein than with its native form. Consequently, they 'pull' the unfolding reaction.[3] Moreover, they may have direct interactions with some standard hydrophobic molecules, e.g., benzene. References
Categories: Solutions | Physical chemistry | Laboratory techniques |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Hofmeister_series". A list of authors is available in Wikipedia. |