To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Nitrogen laserA Nitrogen laser is a gas laser operating in the ultraviolet range (typically 337 nm), using molecular nitrogen as its gain medium, pumped by an electrical discharge. Product highlightGain mediumThe gain medium is nitrogen molecules in the gas phase. The nitrogen laser is a 3-level laser. In contrast to a ruby laser or to other more typical 4-level lasers, the upper laser level of nitrogen is directly pumped, imposing no speed limits on the pump. Pumping is normally provided by direct electron impact; the electrons must have sufficient energy, or they will fail to excite the upper laser level. Typically reported optimum values are in the range of 80 to 100 eV per Torr·cm pressure of nitrogen gas. There is a 40 ns upper limit of laser lifetime at low pressures and the lifetime becomes shorter as the pressure increases. The lifetime is only 1 to 2 ns at 1 atmosphere. In general
For various reasons the efficiency of the nitrogen laser is low, typically 0.1% or less, though nitrogen lasers with efficiency of up to 3% have been reported in the literature. The strongest lines are at 337.1 nm *wavelength in the ultraviolet. Other lines have been reported at 357.6 nm, also ultraviolet. This information refers to the second positive system of molecular nitrogen, which is by far the most common. There are also lines in the far-red and infrared from the first positive system, and a visible blue laser line from the molecular nitrogen positive (1+) ion. The metastable lower level lifetime is 40 μs,[3] thus, the laser self-terminates, typically in less than 20 ns. This type of self-termination is sometimes referred to as “bottlenecking in the lower level”. Thus, in contrast to the helium-neon laser, the nitrogen laser can only operate in pulsed mode. Many nitrogen lasers terminate even more rapidly, but this is usually because the drive circuitry cannot provide sufficiently energetic electrons for more than a few ns. It might be thought that the short pulsewidth of the nitrogen laser is caused by the short lifetime of the upper laser level, but that is demonstrably false. Several organic dyes with upper level lifetimes of less than 10 ns have been used in continuous mode. The upper level lifetime does limit the attainable pulsewidth in the case of the nitrogen laser, but this is caused by the bottlenecking issue and is not an inherent feature of short upper-state lifetime. Repetition rates can range as high as a few kHz, provided adequate gas flow and cooling of the structure are provided. Cold nitrogen is a better medium than hot nitrogen, and this appears to be part of the reason that the pulse energy and power drop as the repetition rate increases to more than a few pulses per second. There are also, apparently, issues involving ions remaining in the laser channel. Typical devicesThe gas pressure in a nitrogen laser ranges from a few mbar to as much as several bar. Air, which is 78% nitrogen, can be used, but more than 0.5% oxygen poisons the laser. Furthermore, air provides significantly less output energy than pure nitrogen or a mixture of nitrogen and helium. The pulse energy ranges from µJ to mJ and a peak power in the range of kW to more than 3 MW can be achieved. The pulse temporal width is between a few hundred picoseconds (typically at 1 atmosphere partial pressure of nitrogen) and a maximum of approximately 30 nanoseconds at reduced pressure (typically some dozens of Torr), though fwhm pulsewidths of 6 to 8 ns are typical. Nitrogen lasers can operate superfluorescently (without a resonator cavity), though it is common to put a mirror at one end such that the output is emitted from the opposite end. The gain medium is usually pumped by a transverse electrical discharge. When the pressure is at (or above) 1013 mbar (atmospheric pressure), the configuration is called a TEA laser Transverse Electrical discharge in gas at Atmospheric pressure. Application
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Nitrogen_laser". A list of authors is available in Wikipedia. |





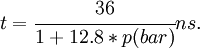 [1],[2]
[1],[2]


