To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Relative humidity
Relative humidity is a term used to describe the amount of water vapor that exists in a gaseous mixture of air and water. Product highlight
DefinitionThe relative humidity of an air-water mixture is defined as the ratio of the partial pressure of water vapor in the mixture to the saturated vapor pressure of water at a given temperature. Relative humidity is expressed as a percentage and is calculated in the following manner:
where:
A common misconceptionOften the concept of air holding water vapor is used in the description of relative humidity. Relative humidity is wholly understood in terms of the physical properties of water alone and therefore is unrelated to this concept. [1] This misconception is likely a result of the use of the word saturation which is often mis-used in definitions of relative humidity. In the present context saturation refers to the saturation state of water vapor [2], not the solubility of one material in another. The thermophysical properties of water-air mixtures encountered at atmospheric conditions can be reasonably approximated by assuming that they behave like a mixture of ideal gases. [3]. For many practical purposes this assumption implies that both components (air and water) behave independently of each other and therefore the physical properties of the mixture can be estimated by considering the physical properties of each component separately. This is reflected in the definition of relative humidity - only the physical properties of water are considered when determining the relative humidity of a mixture. Related conceptsThe term relative humidity is reserved for systems of water vapor in air. The term relative saturation is used to describe the analogous property for systems consisting of a condensable phase other than water or a non-condensable phase other than air. [4] The relative humidity of a system is dependent not only on the temperature but also on the absolute pressure of the system of interest. Therefore, a change in relative humidity can be explained by a change in system temperature, a change in the absolute pressure of the system, or change in both of these system properties. Other important facts
The statement that relative humidity can never be above 100%, while a fairly good guide, is not absolutely accurate, without a more sophisticated definition of humidity than the one given here. An arguable exception is the Wilson cloud chamber which uses, in nuclear physics experiments, an extremely brief state of "supersaturation" to accomplish its function. For a given dewpoint and its corresponding absolute humidity, the relative humidity will change inversely, albeit nonlinearly, with the temperature. This is because the partial pressure of water increases with temperature – the operative principle behind everything from hair dryers to dehumidifiers. Due to the increasing potential for a higher water vapor partial pressure at higher air temperatures, the water content of air at sea level can get as high as 3% by mass at 30 °C (86 °F) compared to no more than about 0.5% by mass at 0 °C (32 °F). This explains the low levels (in the absence of measures to add moisture) of humidity in heated structures during winter, indicated by dry skin, itchy eyes, and persistence of static electric charges. Even with saturation (100% relative humidity) outdoors, heating of infiltrated outside air that comes indoors raises its moisture capacity, which lowers relative humidity and increases evaporation rates from moist surfaces indoors (including human bodies.) Similarly, during summer in humid climates a great deal of liquid water condenses from air cooled in air conditioners. Warmer air is cooled below its dewpoint and the excess water vapor condenses. This phenomenon is the same as that which causes water droplets to form on the outside of a cup containing an ice-cold drink. A useful rule of thumb is that the maximum absolute humidity doubles for every 20 °F (11.1 °C) increase in temperature. Thus, the relative humidity will drop by a factor of 2 for each 20 °F (11.1 °C) increase in temperature, assuming conservation of absolute moisture. For example, in the range of normal temperatures, air at 70 °F (21.1 °C) and 50% relative humidity will become saturated if cooled to 50°F (10 °C), its dewpoint and 40 °F (4.4 °C) air at 80% relative humidity warmed to 70 °F (21.1 °C) will have a relative humidity of only 10% and feel dry. By comparison, a relative humidity between 40% and 60% is considered healthy and comfortable in comfort controlled environments (ASHRAE Standard 55[5]). This latter example leads to a RH of perhaps 30%, not 10%. T rises from 40 °F (4.4 °C) to 60 °F (15.6 °C), RH falls from 80% to 40%, T rises from 60 °F (15.6 °C) to 70 °F (21.1 °C), RH falls perhaps a quarter, from 40% to 30% - not to 10%. Water vapor is a lighter gas than air at the same temperature, so humid air will tend to rise by natural convection. This is a mechanism behind thunderstorms and other weather phenomena. Relative humidity is often mentioned in weather forecasts and reports, as it is an indicator of the likelihood of precipitation, dew, or fog. In hot summer weather, it also increases the apparent temperature to humans (and other animals) by hindering the evaporation of perspiration from the skin as the relative humidity rises. This effect is calculated as the heat index or humidex. A device used to measure humidity is called a hygrometer, one used to regulate it is called a humidistat, or sometimes hygrostat. (These are analogous to a thermometer and thermostat for temperature, respectively.) References
See also
Categories: Psychrometrics | Atmospheric thermodynamics |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Relative_humidity". A list of authors is available in Wikipedia. |





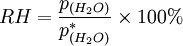
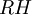 is the relative humidity of the mixture being considered;
is the relative humidity of the mixture being considered;
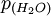 is the partial pressure of water vapor in the mixture; and
is the partial pressure of water vapor in the mixture; and
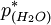 is the
is the 

