To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Sector instrumentA sector instrument is a general term for a class of mass spectrometer that utilizes a static electric or magnetic sector or some combination of the two (separately in space) as a mass analyzer.[1] A popular combination of these sectors has been the BEB (magnetic-electric-magnetic). Most modern sector instruments are double focusing instruments in that they focus the ion beams both in direction and velocity.[2] Product highlight
TheoryThe behavior of ions in a homogeous, linear, static electric or magnetic field (separately) as is found in a sector instrument is simple. The physics are described by a single equation called the Lorentz force law. This equation is the fundamental equation of all mass specrometric techniques and applies in non-linear, non-homogeneous cases too and is an important equation in the field of electrodynamics generally. where E is the electric field strength, B is the magnetic field induction, q is the charge of the particle, v is its current velocity (expressed as a vector), and × is the cross product. So the force on an ion in a linear homogeous electric field (an electric sector) is:
in the direction of the electric field, with positive ions and opposite that with negative ions.
The force is only dependent on the charge and electric field strength. The lighter ions will be deflected more and heavier ions less due to the difference in inertia and the ions will physically separate from each other in space into distinct beams of ions as they exit the electric sector. And the force on an ion in a linear homogeous magnetic field (a magnetic sector) is:
perpendicular to both the magnetic field and the velocity vector of the ion itself, in the direction determined by the right-hand rule of cross products and the sign of the charge. The force in the magnetic sector is complicated by the velocity dependence but with the right conditions (uniform velocity for example) ions of different masses will separate physically in space into different beams as with the electric sector. Classic GeometriesThese are some of the classic geometries from mass spectrographs which are often used to distinguish different types of sector arrangements, although most current instruments do not fit precisely into any of these categories as the designs have evolved further. Bainbridge-JordanThe sector instrument geometry consists of a 127.30° do not experience a resultant force; they pass freely through a slit, and are then subject to another magnetic field, transversing a semi-circular path and striking a photographic plate. The mass of the isotope is determined through subsequent calculation. Mattauch-HerzogThe Mattauch-Herzog geometry consists of a 31.82° ( Nier-JohnsonThe Nier-Johnson geometry consists of a 90° electric sector, a long intermediate drift length and a 60° magnetic sector of the same curvature direction.[5][6] Hinterberger-KonigThe Hinterberger-Konig geometry consists of a 42.43° electric sector, a long intermediate drift length and a 130° magnetic sector of the same curvature direction. TakeshitaThe Takeshita geometry consists of a 54.43° electric sector, and short drift length, a second electric sector of the same curvature direction followed by another drift length before a 180° magnetic sector of opposite curvature direction. MatsudaThe Matsuda geometry consists of a 85° electric sector, a quadrupole lens and a 72.5° magnetic sector of the same curvature direction.[7] See also
References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Sector_instrument". A list of authors is available in Wikipedia. |





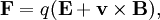
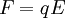 ,
,
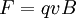 ,
,
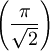 electric sector without an initial drift length followed by a 60° magnetic sector with the same direction of curvature. Sometimes called a "Bainbridge mass spectrometer," this configuration is often used to determine
electric sector without an initial drift length followed by a 60° magnetic sector with the same direction of curvature. Sometimes called a "Bainbridge mass spectrometer," this configuration is often used to determine 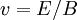
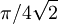 radians) electric sector, a drift length which is followed by a 90° magnetic sector of opposite curvature direction.
radians) electric sector, a drift length which is followed by a 90° magnetic sector of opposite curvature direction.

