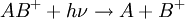To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Tandem mass spectrometryTandem mass spectrometry, also known as MS/MS, involves multiple steps of mass spectrometry selection, with some form of fragmentation occurring in between the stages.[1] Product highlight
Tandem MS instrumentsMultiple stages of mass analysis separation can be accomplished with individual mass spectrometer elements separated in space or in a single mass spectrometer with the MS steps separated in time. In tandem mass spectrometry in space, the separation elements are physically separated and distinct, although there is a connection between the elements to maintain high vacuum. These elements can be sectors, transmission quadrupole, or time-of-flight. In a tandem mass spectrometry in time instrument, the separation is accomplished with ions trapped in the same place, with multiple separation steps taking place over time. A quadrupole ion trap or FTMS instrument can be used for such an analysis. Trapping instruments can perform multiple steps of analysis, which is sometimes referred to as MSn (MS to the n). Often the number of steps, n, is not indicated, but occasionally the value is specified; for example MS3 indicates three stages of separation. NotationFor tandem mass spectrometry in space, the different elements are often noted in shorthand.
The notation can be combined to indicate various hybrid instrument, for example
Tandem MS ExperimentsThere are a number of different tandem MS experiments, which each have their own applications and offer their own information. An instrument equipped for tandem MS can still be used to run MS experiments. Tandem MS can be done in either time or space. Tandem MS in space involves the physical separation of the instrument components (QqQ or QTOF), tandem MS in time involves the use of an ion trap. Tandem MS ModesThere are four main scan experiments possible using MS/MS.
Fragmentation in tandem mass spectrometryFragmentation of gas-phase ions is essential to tandem mass spectrometry and occurs between different stages of mass analysis. There are many methods used to fragment the ions and can result in different types of fragmentation and thus different information about the structure and composition of the molecule. In-source fragmentationOften, the ionization process is sufficiently violent to leave the resulting ions with sufficient internal energy to fragment within the mass spectrometer. If the product ions persist in their non-equilibrium state for a moderate amount of time before auto-dissociation this process is called metastable fragmentation.[3] [4] Nozzle-skimmer fragmentation refers to the purposeful induction of in-source fragmentation by increasing the nozzle-skimmer potential on usually electrospray based instruments. Although in-source fragmentation allows for fragmentation analysis, it is not technically tandem mass spectrometry unless metastable ions are mass analyzed or selected before auto-dissociation and a second stage of analysis is performed on the resulting fragments. In-source fragmentation is often used in addition to tandem mass spectrometry (with post-source fragmentation) to allow for two steps of fragmentation in a pseudo MS3-type of experiment.[5] Post-source fragmentationPost-source fragmentation is most often what is being used in a tandem mass spectrometry experiment. Energy can also be added to the, usually already vibrationally excited, ions through post-source collisions with neutral atoms or molecules, the absorption of radiation, or the transfer or capture of an electron by a multiply charged ion. Collision-induced dissociation (CID), also called collisionally activated dissociation (CAD), involves the collision of an ion with a neutral atom or molecule in the gas phase and subsequent dissociation of the ion.[6][7] For example, consider where the ion AB + collides with the neutral species M and subsequently breaks apart. The details of this process are described by collision theory. If an electron is added to a multiply-charged positive ion, the Coulomb energy is liberated. Adding a free electron is called electron capture dissociation (ECD),[8] and is represented by for a multiply-protonated molecule M. Adding an electron through an ion-ion reaction is called electron transfer dissociation (ETD),[9] and is represented by
The energy required for dissociation can be added by photon absorption, resulting in ion photodissociation and represented by where hν represents the photon absorbed by the ion. Ultraviolet lasers can be used, but can lead to excessive fragmentation of biomolecules.[10] Infrared photons will heat the ions and cause dissociation if enough of them are absorbed. This process is called infrared multiphoton dissociation (IRMPD) and is often accomplished with a carbon dioxide laser and an ion trapping mass spectrometer such as a FTMS.[11] Blackbody radiation can also be used in a technique known as blackbody infrared radiative dissociation (BIRD).[12] In the BIRD method, the entire mass spectrometer vacuum chamber is heated to create infrared radiation. Peptide fragmentationA peptide sequence tag obtained by tandem mass spectrometry can be used to identify a peptide in a protein database.[14][15][16] A notation has been developed for indicating peptide fragments that arise from a tandem mass spectrum.[13] Peptide fragment ions are indicated by a, b, or c if the charge is retained on the N-terminus and by x, y or z if the charge is maintained on the C-terminus. The subscript indicates the number of amino acid residues in the fragment. Superscripts are sometimes used to indicate neutral losses in addition to the backbone fragmentation, * for loss of ammonia and ° for loss of water. Although peptide backbone cleavage is the most useful for sequencing and peptide identification other fragment ions may be observed under certain conditions. These include the side chain loss ions d, v, w and immonium ions. [17][18] Oligosaccharide fragmentationOligosaccharides may be sequenced using tandem mass spectrometry in a similar manner to peptide sequencing. Fragmentation generally occurs on either side of the glycosidic bond (b, c, y and z ions) but also under more energetic conditions through the sugar ring structure in a cross-ring cleavage (x ions). Again trailing subscripts are used to indicate position of the cleavage along the chain. For cross ring cleavage ions the nature of the cross ring cleavage is indicated by preceding superscripts.[19][20] See also
References
Bibliography
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Tandem_mass_spectrometry". A list of authors is available in Wikipedia. |





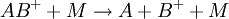
![[M + nH]^{n+} + e^- \to \bigg[ [M + (n-1)H]^{(n-1)+} \bigg]^* \to fragments](images/math/b/e/a/bea5d60c0444fbf8c6c739d50412db50.png)
![[M + nH]^{n+} + A^- \to \bigg[ [M + (n-1)H]^{(n-1)+} \bigg]^* + A \to fragments](images/math/b/e/d/bed9e5f4a6b641eaec1b272b7427f65a.png) .
.