To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Sodium azide
Sodium azide is the chemical compound with the formula NaN3. This colourless salt is a common reagent in organic synthesis, and it is a component in many car airbag systems. Product highlight
Structure, synthesis, and principal reactionsStructureSodium azide is ionic. The N3− group is centrosymmetric with N-N distances of 1.18 Å.[1] Sodium azide is highly soluble in water. Such solutions contain minute amounts of hydrogen azide, as described by the following equilibrium:
PreparationThe common synthesis method is the "Wislicenus process," which proceeds in two steps from ammonia. In the first step, ammonia is converted to sodium amide:
The sodium amide is subsequently combined with nitrous oxide.
Alternatively the salt can be obtained by the reaction of sodium nitrate with sodium amide.[2] ReactionsTreatment of sodium azide with strong acids gives the corresponding acid, hydrazoic acid:
Sodium azide cannot be melted, but decomposes vigorously to sodium metal and nitrogen gas at approximately 300 °C. An electrical charge triggered by automobile impact heats the salt to explosively release nitrogen gas inside the airbag:
The sodium that is formed is a potential hazard itself and, in automobile airbags, it is converted by reaction with other ingredients, such as potassium nitrate (KNO3) and silica (SiO2), into an inert alkaline silicate 'glass'.[3] Sodium azide is used in organic synthesis to introduce the azide functional group by displacement of a halide.[4] The azide functional group can thereafter be converted to an amine by reduction with either lithium aluminum hydride, or a tertiary phosphine such as triphenylphosphine in the Staudinger reaction. Sodium azide can be destroyed by treatment with acidic sodium nitrite solution:[5]
Biochemistry and biomedical usesSodium azide is a useful probe reagent, mutagen, and preservative. In hospitals and laboratories, it is a biocide; it is especially important in bulk reagents and stock solutions which may otherwise support bacterial growth where the sodium azide acts as a bacteriostatic by inhibiting cytochrome oxidase in gram-negative bacteria; gram-positive (streptococci, pneumococci, lactobacilli) are resistant[6] (a characteristic similar to antibiotic resistance). It is also used in agriculture (farming) for pest control. Azide inhibits cytochrome oxidase by binding irreversibly to the heme cofactor in a process similar to the action of carbon monoxide. Sodium azide is particularly affects organs that undergo high rates of respiration, such as the heart and the brain. Toxic effectsSodium azide is often comparable with cyanide, as they give similar symptoms.[citation needed] Exposure to sodium azide has some or all of the following symptoms within minutes: rapid breathing, restlessness, dizziness, weakness, headache, nausea and vomiting, rapid heart rate, red eyes (gas or dust exposure), clear drainage from the nose (gas or dust exposure), cough (gas or dust exposure), skin burns and blisters (explosion or direct skin contact. Exposure to a large amount of sodium azide may cause these other health effects as well: convulsions, low blood pressure, low heart rate, loss of consciousness, and lung injury, respiratory failure leading to death. References
Categories: Sodium compounds | Azides | Explosive chemicals |
|||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Sodium_azide". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||||||||||||||||





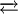 HN3 K = 10−4.6
HN3 K = 10−4.6


