To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Vapour Pressure DeficitVapour Pressure Deficit, or VPD, is the difference (deficit) between the amount of moisture in the air and how much moisture the air can hold when it is saturated. Once air becomes saturated water will condense out to form clouds, dew or films of water over leaves. It is this last instance that makes VPD important for greenhouse regulation. If a film of water forms on a plant leaf it becomes far more susceptible to rot. On the other hand, as the VPD increases the plant needs to draw more water from its roots (and if it is a cutting, dry out and die). For this reason the ideal range for VPD in a greenhouse is from 0.45 kPa to 1.25 kPa, ideally sitting at around 0.85 kPA. As a general rule, most plants grow well at VPDs of between 0.8 to 0.95 kPa [1] Product highlightComputing VPD for plants in a greenhouseTo compute the VPD[2] we need the ambient (greenhouse) air temperature, the relative humidity and if possible, the canopy air temperature. We must then compute the saturation pressure. Saturation pressure can be looked up in a psychrometric chart or derived from the Arrhenius equation, a way to compute it directly from temperature is
where A = − 1.88x104 B = − 13.1 C = − 1.5x10 − 2 D = 8x10 − 7 E = − 1.69x10 − 11 F = 6.456 T – Temperature of the air in K, T(K) = T(°C) + 273.15 We compute this pressure for both the ambient and canopy temperatures. We then can compute the actual partial pressure of the water vapour in the air by multiplying by the relative humidity: vpair = vpsat * relative humidity and finally VPD using vpsat − vpair or vpcanopy sat − vpair when the canopy temperature is known. References |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Vapour_Pressure_Deficit". A list of authors is available in Wikipedia. |





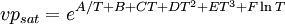 kPa
kPa


