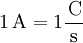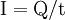To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Ampere
The ampere, in practice often shortened to amp, (symbol: A) is a unit of electric current, or amount of electric charge per second. The ampere is an SI base unit, and is named after André-Marie Ampère, one of the main discoverers of electromagnetism. Product highlight
DefinitionThe ampere is a constant current which, if maintained in two straight parallel conductors of infinite length, of negligible circular cross section, and placed 1 metre apart in a vacuum, would produce between these conductors a force equal to 2×10–7 newton per metre of length.[1] The ampere is a base unit, along with the metre, kelvin, second, mole, candela and the kilogram: it is defined without reference to the quantity of electric charge. The unit of charge, the coulomb, is defined, as a derived unit, to be the amount of charge displaced by a one ampere current in the time of one second. As a result, electric current is also the time rate of change or displacement of electric charge. One ampere represents the rate of 1 coulomb of charge per second. ExplanationBecause it is a base unit, the definition of the ampere is not tied to any other electrical unit. The definition for the ampere is equivalent to fixing a value of the permeability of vacuum to μ0 = 4π×10−7 H/m. Prior to 1948, the so-called "international ampere" was used, defined in terms of the electrolytic deposition rate of silver.[2] The older unit is equal to 0.999 85 A. The ampere is most accurately realized using a watt balance, but is in practice maintained via Ohm's Law from the units of EMF and resistance, the volt and the ohm, since the latter two can be tied to physical phenomena that are relatively easy to reproduce, the Josephson junction and the quantum Hall effect, respectively. The unit of electric charge, the coulomb, is defined in terms of the ampere: one coulomb is the amount of electric charge (formerly quantity of electricity) carried in a current of one ampere flowing for one second.[3] Current, then, is the rate at which charge flows through a wire or surface. One ampere of current (I) is equal to a flow of one coulomb of charge (Q) per second of time (t): Proposed future definitionSince a coulomb is approximately equal to 6.24150948×1018 elementary charges, one ampere is approximately equivalent to 6.24150948×1018 elementary charges, such as electrons, moving past a boundary in one second. As with other SI base units, there have been proposals to redefine the kilogram in such a way as to define some presently measured physical constants to fixed values. One proposed definition of the kilogram is:
This redefinition of the kilogram has the effect of fixing the elementary charge to be e = 1.60217653×10-19 C and would result in a functionally equivalent definition for the coulomb as being the sum of exactly 6 241 509 479 607 717 888 elementary charges and the ampere as being the electrical current of exactly 6 241 509 479 607 717 888 elementary charges per second. This is consistent with the current 2002 CODATA value for the elementary charge which is 1.60217653×10-19 ± 0.00000014×10-19 C. CIPM recommendationInternational Committee for Weights and Measures (CIPM) Recommendation 1 (CI-2005): Preparative steps towards new definitions of the kilogram, the ampere, the kelvin and the mole in terms of fundamental constants The International Committee for Weights and Measures (CIPM),
See also
References
|
|||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Ampere". A list of authors is available in Wikipedia. |