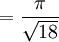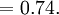To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Atomic packing factorIn crystallography, atomic packing factor or packing fraction is the fraction of volume in a crystal structure that is occupied by atoms. It is dimensionless and always less than unity. For practical purposes, the APF of a crystal structure is determined by assuming that atoms are rigid spheres. It is represented mathematically by Product highlightwhere N is the number of atoms in the crystal and V is the volume. It can be proven mathematically that one-component (one type of atom) close-packed structures, those that have the most dense arrangement of atoms, has an APF of 0.74. In reality, this number can be higher given specific intermolecular factors. For multiple-component structures, the APF can exceed 0.74. Worked exampleThe body-centered cubic crystal structure contains eight atoms on each corner of the cube and one atom in the center. Because the volume of the corner atoms are shared between adjacent cells, each BCC crystal only contains two whole atoms. Each corner atom touches the center atom. A line that is drawn from one corner of the cube through the center and to the other corner passes through 4r, where r is the radius of an atom. By geometry, the length of the diagonal is a*√3. Therefore, the length of each side of the BCC structure can be related to the radius of the atom by Knowing this and the formula for the volume of a sphere, it becomes possible to calculate the APF.
Knowing this and the formula for the volume of a sphere, it becomes possible to calculate the APF.
APF of common structuresBy identical procedures, the ideal atomic packing factors of all crystal structures can be found. The common ones are collected here as reference.
References
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Atomic_packing_factor". A list of authors is available in Wikipedia. |


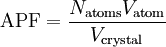



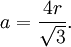
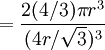
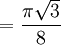
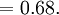
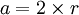
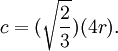
(c)}](images/math/a/a/d/aad65b147621e2b4f42bb4a65be83898.png)
^2(\sqrt{\frac{2}{3}})(4r)}](images/math/a/0/2/a02f062527011e27a5cf78667f235b01.png)
(16r^3)}](images/math/0/b/b/0bb7ba56a4a21979bcc7226b51efc428.png)