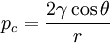To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Capillary pressureIn fluid statics, capillary pressure is the difference in pressure across the interface between two immiscible fluids. The pressure difference is proportional to the surface tension, γ, and inversely proportional to the effective radius, r, of the interface, it also depends on the wetting angle, θ, of the liquid on the surface of the capillary. Product highlightThe capillary pressure pcis defined as
for the case of one phase being gas with pressure pg and the other phase being water with pressure pw. The equation for capillary pressure is only valid under capillary equilibrium, which means that there can not be any flowing phases. See also
References
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Capillary_pressure". A list of authors is available in Wikipedia. |