To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Characteristic state function
The characteristic state function in statistical mechanics refers to a particular relationship between the partition function of an ensemble. Product highlightIn particular, if the partition function P satisfies
in which Q is a thermodynamic quantity, then Q is known as the "characteristic state function" of the ensemble corresponding to "P". Beta refers to the thermodynamic beta. Examples
|
||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Characteristic_state_function". A list of authors is available in Wikipedia. |





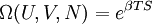 hence, its characteristic state function is
hence, its characteristic state function is 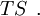 This quantity roughly speaking, denotes the energy of the entropy at a particular temperature.
This quantity roughly speaking, denotes the energy of the entropy at a particular temperature.
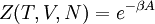 hence, its characteristic state function is the
hence, its characteristic state function is the 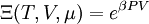 , so its characteristic state function is the total
, so its characteristic state function is the total 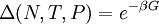 so its characteristic function is the
so its characteristic function is the 

