To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Combinatorial chemistryCombinatorial chemistry (computer synthesis) involves the rapid synthesis or the computer simulation of a large number of different but structurally related molecules. Product highlight
IntroductionSynthesis of molecules in a combinatorial fashion can quickly lead to large numbers of molecules. For example, a molecule with three points of diversity (R1, R2, and R3) can generate Although combinatorial chemistry has only really been taken up by industry since the 1990s, its roots can be seen as far back as the 1960s when a researcher at Rockefeller University, Bruce Merrifield, started investigating the solid-phase synthesis of peptides. In the 1980s researcher H. Mario Geysen developed this technique further, creating arrays of different peptides on separate supports. In its modern form, combinatorial chemistry has probably had its biggest impact in the pharmaceutical industry. Researchers attempting to optimize the activity profile of a compound create a 'library' of many different but related compounds. Advances in robotics have led to an industrial approach to combinatorial synthesis, enabling companies to routinely produce over 100,000 new and unique compounds per year (see medicinal chemistry). In order to handle the vast number of structural possibilities, researchers often create a 'virtual library', a computational enumeration of all possible structures of a given pharmacophore with all available reactants. Such a library can consist of thousands to millions of 'virtual' compounds. The researcher will select a subset of the 'virtual library' for actual synthesis, based upon various calculations and criteria (see ADME, computational chemistry, and QSAR). Materials science has applied to the techniques of combinatorial chemistry to the discovery of new materials. This work was pioneered by P.G. Schultz et al. in the mid nineties (Science, 1995, 268: 1738-1740) in the context of luminescent materials obtained by co-deposition of elements on a silicon substrate. Work has been continued by several academic groups as well as companies with large research and development programs (Symyx Technologies, GE, etc). Diversity-oriented librariesEven though combinatorial chemistry has been an essential part of early drug discovery for more than two decades, so far only one de novo combinatorail chemistry-synthesized chemical has been approved for clinical use by FDA (a multikinase inhibitor indicated for advanced renal cancer). (newman & Cragg 2007) The analysis of poor success rate of the approach has been suggested to connect with the rather limited chemical space covered by products of combinatorial chemistry. When comparing the properties of compounds in combinatorial chemistry libraries to those of approved drugs and natural products, Feher and Schmidt (2003) noted that combinatorial chemistry libraries suffer particularly from the lack of chirality, as well as structure rigidity, both of which are widely regarded as drug-like properties. Even though natural product drug discovery has not probably been the most fashionable trend in pharmaceutical industry in recent times, a large proportion of new chemical entities still is nature-derived compounds, and thus, it has been suggested that effectiveness of combinatorial chemistry could be improved by enhancing the chemical diversity of screening libraries. As chirality and rigidity are the two most important features distinguishing approved drugs and natural products from compounds in combinatorial chemistry libraries, these are the two issues emphasized in so-called diversity oriented libraries, i.e. compound collections that aim at coverage of the chemical space, instead of just huge number of compounds. TriviaIn the 8th edition of the International Patent Classification (IPC), which entered into force on January 1, 2006, a special subclass has been created for patent applications and patents related to inventions in the domain of combinatorial chemistry: "C40B". See also
References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Combinatorial_chemistry". A list of authors is available in Wikipedia. |





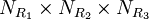 possible structures, where
possible structures, where 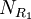 ,
, 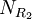 , and
, and 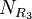 are the number of different substituents utilized.
are the number of different substituents utilized.


