To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
DART ion sourceA DART ion source (Direct analysis in real time) refers to an atmospheric-pressure ion source for mass spectrometry or ion mobility spectrometry that permits analysis of gases, liquids, solids, or materials on surfaces in open air at ground potential under ambient conditions.[1] Samples such as pills, clothing, human skin, plant material, liquid droplets, etc. are held in front of the DART source resulting in immediate formation of ions that are directed into a mass spectrometer or ion mobility spectrometer for rapid analysis. DART was first conceived in 2001, constructed in early 2003, and publicly announced concurrent with the introduction of a commercial product in 2005. The acronym DART stands for “Direct Analysis in Real Time”. Samples exposed to the DART gas stream will rapidly generate ions that are carried by the gas stream into the sampling orifice of the mass spectrometer atmospheric pressure interface. The gas stream can graze a sample surface or be reflected off the sample surface. Liquids can be sampled by dipping an object (such as a glass rod) into the liquid sample to be measured. Vapors are introduced directly into the DART gas stream. Product highlight
Principle
The DART source operates by exposing the sample to a dry gas stream (typically helium or nitrogen) that contains long-lived electronically or vibronically excited neutral atoms or molecules (or "metastables"). Excited states are typically formed in the DART source by creating a glow discharge in a chamber through which the gas flows. Potentials applied to electrostatic lenses remove charged particles from the gas stream. A grid at the exit of the DART source acts as a source of electrons and reduces positive-ion/negative-ion recombination. The excited-state species can interact directly with the sample to desorb and ionize the sample. This process is referred to as Penning ionization, a reaction between an excited-state neutral atom or molecule M* and a substrate S that has an ionization potential with a lower energy than the internal energy of the excited-state species, resulting in the formation of a substrate radical molecular cation S+. and an electron e-: The helium 23S state has an internal energy of 19.8 electron volts, which is sufficient to ionize most organic molecules. Alternatively, the excited-state species can interact with atmospheric gases such as water and oxygen to form reagent ions that undergo chemical ionization reactions that result in ionization of the analyte. In negative-ion mode, electrons e- are thermalized by collisions with gas molecules, G. Atmospheric oxygen captures an electron and reacts with the sample to produce negative ions. Exposure to excited-state species assists in desorbing materials from surfaces. The DART gas stream can also be heated to enhance desorption and/or to decompose large molecules (such as industrial polymers) to produce characteristic fragments that can be used for identification. DART produces relatively simple mass spectra, dominated by protonated molecules [M+H]+ in positive-ion mode, or deprotonated molecules [M-H]- in negative-ion mode. Depending on the nature of the molecule, other species may be formed, such as M+. from polynuclear aromatic hydrocarbons. Fragmentation may occasionally be observed for some molecules. Multiple-charge ions and alkali metal cation aducts are never observed, but addition of ammonia or other "dopants" to the DART gas stream can be used to form single-charge adducts such as [M+NH4]+ or [M+Cl]- for compounds that would not readily form molecular ions or protonated molecules. For example, the explosives nitroglycerin and HMX do not form [M-H]-,but readily form [M+Cl]- if chloride is present. ApplicationsDART is useful for small-molecule analysis. It is not a technique for analysis of large biomolecules such as proteins, although industrial polymers and some large molecules such as cellulose can be fragmented to produce characteristic "fingerprint" patterns. DART's relative insensitivity to contamination and lack of carryover between samples makes it possible to analyze many materials with little or no sample preparation. It should be noted that the major limitation for direct analysis is the ability to distinguish the sample from other environmental compounds. High-resolution mass spectrometry and/or tandem mass spectrometry may be required to identify a target compound in the presence of interferences. Most DART applications to date have been measured with a time-of-flight mass spectrometer.
DART has found a variety of applications including forensics and security, chemical synthesis (reaction monitoring and characterization of synthetic products), analysis of foods and beverages, environmental analysis, and art conservation. See alsoNotes
References
Manufacturers of DART ion sources
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "DART_ion_source". A list of authors is available in Wikipedia. |





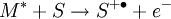
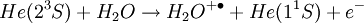
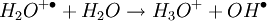
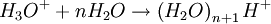
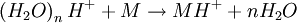
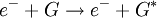
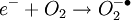
![O{_2^{-\bullet}} + S \to [S - H]^- + OOH^\bullet](images/math/8/f/a/8fa2c02ab58f4c98ee54cf835bfe039f.png)
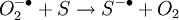
![O{_2^{-\bullet}} + S \to \left[S+O_2\right]^{-\bullet *} + G \to \left[S+O_2\right]^{-\bullet } + G^*](images/math/5/0/b/50ba4e1679559ed71474d880ae3eac32.png)


