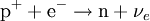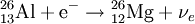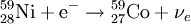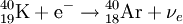To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Electron captureElectron capture (sometimes called Inverse Beta Decay) is a decay mode for isotopes that will occur when there are too many protons in the nucleus of an atom and insufficient energy to emit a positron; however, it continues to be a viable decay mode for radioactive isotopes that can decay by positron emission. If the energy difference between the parent atom and the daughter atom is less than 1.022 MeV, positron emission is forbidden and electron capture is the sole decay mode. For example, Rubidium-83 will decay to Krypton-83 solely by electron capture (the energy difference is about 0.9 MeV). Product highlightIn this case, one of the orbital electrons, usually from the K or L electron shell (K-electron capture, also K-capture, or L-electron capture, L-capture), is captured by a proton in the nucleus, forming a neutron and a neutrino. Since the proton is changed to a neutron, the number of neutrons increases by 1, the number of protons decreases by 1, and the atomic mass number remains unchanged. By changing the number of protons, electron capture transforms the nuclide into a new element. The atom moves into an excited state with the inner shell missing an electron. When transiting to the ground state, the atom will emit an X-ray photon (a type of electromagnetic radiation) and/or Auger electrons. examples: (Please note that it is one of the initial atom's own electrons that is captured, not a new, incoming electron as might be suggested by the way the above reactions are written.) Radioactive isotopes which decay by pure electron capture can, in theory, be inhibited from radioactive decay if they are fully ionized ("stripped" is sometimes used to describe such ions). It is hypothesized that such elements, if formed by the r-process in exploding supernovae, are ejected fully ionized and so do not undergo radioactive decay as long as they do not encounter electrons in outer space. Anomalies in elemental distributions are thought to be partly a result of this effect on electron capture. Chemical bonds can also affect the rate of electron capture to a small degree (generally less than 1%) depending on the proximity of electrons to the nucleus.[1] Around the elements in the middle of the periodic table, isotopes that are lighter than stable isotopes of the same element tend to decay through electron capture, while isotopes heavier than the stable ones decay with a process called negative beta decay. A good example of this effect would be silver, as its light isotopes use electron capture and the heavier ones decay by negative beta emission.
Categories: Nuclear chemistry | Radioactivity |
|||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Electron_capture". A list of authors is available in Wikipedia. | |||||||||||