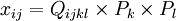To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
ElectrostrictionElectrostriction is a property of all electrical non-conductors, or dielectrics, that causes them to change their shape under the application of an electric field. (Compare with magnetostriction.) Product highlight
ExplanationElectrostriction is a property of all dielectric materials, and is caused by the presence of randomly-aligned electrical domains within the material. When an electric field is applied to the dielectric, the opposite sides of the domains become differently charged and attract each other, reducing material thickness in the direction of the applied field (and increasing thickness in the orthogonal directions due to Poisson's ratio). The resulting strain (ratio of deformation to the original dimension) is proportional to the square of the polarization. Reversal of the electric field does not reverse the direction of the deformation. More formally, the electrostriction coefficient is a fourth order tensor (Qijlk), relating second order strain (xij) and first order polarization tensors (Pk, Pl).
It should be noted that the related piezoelectric effect occurs only in a particular class of dielectrics. Electrostriction is a quadratic effect, unlike piezoelecticity, which is a linear effect. In addition, unlike piezoelectricity, electrostriction cannot be reversed: deformation will not induce an electric field. MaterialsAlthough all dielectrics exhibit some electrostriction, certain engineered ceramics, known as relaxor ferroelectrics, have extraordinarily high electrostrictive constants. The most commonly used are:
See alsoReferences
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Electrostriction". A list of authors is available in Wikipedia. |