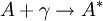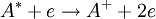To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Hollow cathode lampA hollow cathode lamp (HCL) is type of lamp used in physics and chemistry as a spectral line source and as a frequency tuner for light sources such as lasers. Product highlight
An HCL usually consists of a glass tube containing a cathode made of the material of interest, an anode, and a buffer gas (usually a noble gas). A large voltage across the anode and cathode will cause the buffer gas to ionize, creating a plasma. The buffer gas ions will then be accelerated into the cathode, sputtering off atoms from the cathode. The sputtered cathode atoms will in turn be excited by collisions with other atoms/particles in the plasma. As these excited atoms decay to lower states, they will emit photons, which can then be detected and a spectrum can be determined. An HCL can also be used to tune light sources to a specific atomic transition by making use of the opto-galvanic effect, which is a result of direct or indirect photoionization. By shining the light source into the HCL, one can excite or even eject electrons (directly photoionize) from the atoms inside the lamp, so long as the light source includes frequencies corresponding to the right atomic transitions. Indirect photoionization can then occur when electron collisions with the excited atom eject an atomic electron. The newly created ions cause an increase in the current across the cathode/anode and a resulting change in the voltage, which can then be measured. To tune the light source to a specific transition frequency, a tuning parameter (often the driving current) of the light source is varied. By looking for a resonance on a data plot of the voltage signal versus source tuning parameter, the light source can be tuned to the desired frequency. This is often aided by use of a lock-in circuit. See also
|
||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Hollow_cathode_lamp". A list of authors is available in Wikipedia. | ||||||||||||||||