To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Hypochlorous acid
Chloric(I) acid is a weak acid with the chemical formula HOCl. It forms when chlorine dissolves in water. It cannot be isolated in pure form due to rapid equilibration with its precursor (see below). HOCl is used as a bleach, an oxidizer, a deodorant, and a disinfectant. Product highlight
FormationAddition of chlorine to water gives both chloric(I) acid and hydrochloric acid[2] (HCl):
UsesIn organic synthesis, HOCl converts alkenes to chlorohydrins.[3] In biology, hypochlorous acid is generated in activated neutrophils by myeloperoxidase mediated peroxidation of chloride ions, and contributes to the destruction of bacteria.[4][5][6] Hypochlorous acid is the active sanitizer in hypochlorite based swimming pool products. Chemical reactionsIn aqueous solution, hypochlorous acid partially dissociates into the anion hypochlorite ClO-:
Salts of hypochlorous acid are also called hypochlorites. One of the best known hypochlorites is NaClO, the active ingredient in bleach. In the presence of sunlight, hypochlorous acid decomposes into hydrochloric acid and oxygen, so this reaction is sometimes seen as:
HOCl is considered to be a stronger oxidant than chlorine. Reactivity of HOCl with biomoleculesHypochlorous acid reacts with a wide variety of biomolecules including DNA, RNA,[6][7][8][9] fatty acid groups, cholesterol[10][11][12][13][14][15][16][17] and proteins.[2][18][13][19][20][21][22] Reaction with protein sulfhydral groupsKnox et al.[20] first noted that HOCl was a sulfhydral inhibitor that in sufficient quantity could completely inactivate proteins containing sulfhydral groups. This is because, HOCl oxidises sulfhydral groups leading to the formation of disulfide bonds[23] that can result in crosslinking of proteins. The HOCl mechanism of sulfhydral oxidation is similar to that of chloramine, and may only be bacteriostatic, because once the residual chlorine is dissipated, some sulfhydral function can be restored.[19] One sulfhydral containing amino acid can scavenge up to four molecules of HOCl.[22] Consistent with this, it has been proposed that sulfhydral groups of sulfur containing amino acids can be oxidized a total of three times by three HOCl molecules, with the fourth reacting with the α-amino group. The first reaction yields sulfenic acid (R-SOH) then sulfinic acid (R-SO2H) and finally R-SO3H. Each of those intermediates can also condense with another sulfhydral group causing cross linking and aggregation of proteins. Sulfinic acid and R-SO3H derivatives are only produced at high molar excesses of HOCl, and disulfides are primarily formed at bacteriocidal levels.[9] Disulfide bonds can also be oxidized by HOCl to sulfinic acid.[23] Because the oxidation of sulfhydrals and disulfides evolves hydrochloric acid,[9] this process results in the depletion HOCl. Reaction with protein amino groupsHypochlorous acid reacts readily with amino acids that have amino group side chains, with the chlorine from HOCl displacing a hydrogen resulting in an organic chloramine.[24] Chlorinated amino acids rapidly decompose but protein chloramines are longer lived and retain some oxidative capacity.[22][5] Thomas et al.[5] concluded from their results that most organic chloramines decayed by internal rearrangement and that fewer available NH2 groups promoted attack on the peptide bond resulting in cleavage of the protein. McKenna and Davies[25] found that 10 mM or greater HOCl was necessary to fragment proteins in vivo. Consistent with these results it was later proposed that the chloramine undergoes a molecular rearrangement releasing HCl and ammonia to form an amide.[26] The amide group can further react with another amino group to form a Schiff base causing cross linking and aggregation of proteins.[13] Reaction with DNA and NucleotidesHypochlourous acid reacts slowly with DNA and RNA as well as all nucleotides in vitro.[7][27] GMP is the most reactive because HOCl reacts with both the heterocyclic NH group and the amino group. Similarly TMP with only a heterocyclic NH group that is reactive with HOCl is the second most reactive. AMP and CMP which only have a slowly reactive amino group are less reactive with HOCl.[27] UMP has been reported to be reactive only at a very slow rate.[6][7] The heterocyclic NH groups are more reactive than amino groups and their secondary chloramines are able to donate the chlorine.[9] These reactions likely interfere with DNA base pairing and consistent with this, Prütz[27] has reported a decrease in viscosity of DNA exposed to HOCl similar to that seen with heat denaturation. The sugar moieties are unreactive and the DNA backbone is not broken.[27] NADH can react with chlorinated TMP and UMP as well as HOCl. This reaction can regenerate UMP and TMP and results in the 5-hydroxy derivative of NADH. The reaction with TMP or UMP is slowly reversible to regenerate HOCl. A second slower reaction that results in cleavage of the pyridine ring occurs when excess HOCl is present. NAD+ is inert to HOCl.[27][9] Reaction with lipidsHypochlorous acid reacts with unsaturated bonds in lipids, but not saturated bonds, and the OCl− ion does not participate in this reaction. This reaction occurs by hydrolysis with addition of chlorine to one of the carbons and a hydroxyl to the other. The resulting compound is a chlorhydrin.[10] The polar chlorine disrupts lipid bilayers and could increase permeability.[11] When chlorhydrin formation occurs in lipid bilayers of red blood cells, increased permeability occurs. Disruption could occur if enough chlorhydrin is formed.[10][16] The addition of preformed chlorhydrins to red blood cells can affect permeability as well.[12] Cholesterol chlorhydrins have also been observed,[11][14] but do not greatly affect permeability, and it is believed that Cl2 is responsible for this reaction.[14] Mode of disinfectant actionEscherichia coli exposed to hypochlorous acid lose viability in less than 100 ms due to inactivation of many vital systems.[2][28][29][30][31] Hypochlorous acid has a reported LD50 of 0.0104 ppm - 0.156 ppm[32] and 2.6 ppm caused 100% growth inhibition in 5 minutes.[25] However it should be noted that the concentration required for bactericidal activity is also highly dependent on bacterial concentration.[20] Inhibition of glucose oxidationIn 1948, Knox et al.[20] proposed the idea that inhibition of glucose oxidation was a major factor in the bacteriocidal nature of chlorine solutions. He proposed that the active agent or agents diffused across the cytoplasmic membrane to inactivate key sulfhydral containing enzymes in the glycolytic pathway. This group was also the first to note that chlorine solutions (HOCl) inhibited sulfhydral enzymes. Later studies have shown that at bacteriocidal levels, the cytosol components do not react with HOCl.[1] In agreement with this, McFeters and Camper[33] found that aldolase, an enzyme that Knox et al.[20] proposes would be inactivated, was unaffected by HOCl in vivo. It has been further shown that loss of sulfhydrals does not correlate with inactivation.[19] That leaves the question what causes inhibition of glucose oxidation. The discovery that HOCl blocks induction of β-galactosidase by added lactose[34] led to a possible answer to this question. The uptake of radiolabeled substrates by both ATP hydrolysis and proton co-transport may be blocked by exposure to HOCl preceding loss of viability.[1] From this observation it proposed that HOCl blocks uptake of nutrients by inactivating transport proteins.[1][18][35][33] The question of loss of glucose oxidation has been further explored in terms of loss of respiration. Venkobachar et al.[36] found that succinic dehydrogenase was inhibited in vitro by HOCl and this led to the investigation of the possibility that disruption of electron transport could be the cause of bacterial inactivation. Albrich et al.[6] subsequently found that HOCl destroys cytochromes and iron-sulfur clusters and observed that oxygen uptake is abolished by HOCl and adenine nucleotides are lost. Also observed was, that irreversible oxidation of cytochromes paralleled the loss of respiratory activity. One way of addressing the loss of oxygen uptake was by studying the effects of HOCl on succinate dependent electron transport.[37] Rosen et al.[31] found that levels of reductable cytochromes in HOCl treated cells were normal, and these cells were unable to reduce them. Succinate dehydrogenase was also inhibited by HOCl, stopping the flow of electrons to oxygen. Later studies[29] revealed that Ubiquinol oxidase activity ceases first, and the still active cytochromes reduce the remaining quinone. The cytochromes then pass the electrons to oxygen, which explains why the cytochromes cannot be reoxidized as observed by Rosen et al.[31] However, this line of inquiry was ended when Albrich et al.[2] found that cellular inactivation precedes loss of respiration by using a flow mixing system that allowed evaluation of viability on much smaller time scales. This group found that cells capable of respiring could not divide after exposure to HOCl. Depletion of adenine nucleotidesHaving eliminated loss of respiration Albrich et al.[2] proposes that the cause of death may be due to metabolic dysfunction caused by depletion of adenine nucleotides. Barrette et al.[34] studied the loss of adenine nucleotides by studying the energy charge of HOCl exposed cells and found that cells exposed to HOCl were unable to step up their energy charge after addition of nutrients. The conclusion was that exposed cells have lost the ability to regulate their adenylate pool, based on the fact that metabolite uptake was only 45% deficient after exposure to HOCl and the observation that HOCl causes intracellular ATP hydrolysis. Also confirmed was; that at bacteriocidal levels of HOCl, cytosolic components are unaffected. So it was proposed that modification of some membrane bound protein results in extensive ATP hydrolysis, and this, coupled with the cells inability to remove AMP from the cytosol depresses metabolic function. One protein involved in loss of ability to regenerate ATP has been found to be ATP synthetase.[18] Much of this research on respiration reconfirms the observation that relevant bacteriocidal reactions take place at the cell membrane.[34][18][38] Inhibition of DNA replicationRecently it has been proposed that bacterial inactivation by HOCl is the result of inhibition of DNA replication. When bacteria are exposed to HOCl there is a precipitous decline in DNA synthesis that precedes inhibition of protein synthesis, and closely parallels loss of viability.[25][39] During bacterial genome replication, the origin of replication (oriC in E. coli) binds to proteins that are associated with the cell membrane, and it was observed that HOCl treatment decreases the affinity of extracted membranes for oriC, and this decreased affinity also parallels loss of viability. A study by Rosen et al[40] compared the rate of HOCl inhibition of DNA replication of plasmids with different replication origins and found that certain plasmids exhibited a delay in the inhibition of replication when compared to plasmids containing oriC. Rosen’s group proposed that inactivation of membrane proteins involved in DNA replication are the mechanism of action of HOCl. SafetyHOCl is a strong oxidizer and can form explosive mixtures. Categories: Hypochlorites | Acids | Disinfectants |
|||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Hypochlorous_acid". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||||||||||||||||||||||





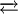 OCl- + H+
OCl- + H+


