To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Mayo-Lewis equationThe Mayo-Lewis equation or copolymer equation in polymer chemistry describes the distribution of monomers in a copolymer [1]: Taking into consideration a monomer mix of two components and with reactivity ratio's defined as: the copolymer equation is given as: with the concentration of the components given in square brackets. The equation gives the copolymer composition at any instant during the polymerization. Product highlight
Limiting casesFrom this equation several limiting cases can be derived:
An example is maleic anhydride and stilbene, with reactivity ratio:
Both of these compounds do not homopolymerize and instead, they react together to give exclusively alternating copolymer. Another form of the equation is:
where
and
When the copolymer composition has the same composition as the feed, this composition is called the azeotrope. Calculation of reactivity ratio'sThe reactivity ratio's can be obtained by rewriting the copolymer equation to:
with
and
A number of copolymerization experiments are conducted with varying monomer ratio's and the copolymer composition is analysed at low conversion. A plot of A semi-empirical method for the determination of reactivity ratio's is called the Q-e scheme. ProofMonomer 1 is consumed with reaction rate [2]:
with Likewise the rate of disappearance for monomer 2 is:
Division of both equations yields:
The ratio of active center concentrations can be found assuming steady state with:
meaning that the concentration of active centres remains constant, the rate of formation for active center of monomer 1 is equal to the rate of their destruction or:
or
References
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Mayo-Lewis_equation". A list of authors is available in Wikipedia. |


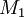 and
and 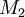 and the four different reactions that can take place at the reactive chain end terminating in either monomer (
and the four different reactions that can take place at the reactive chain end terminating in either monomer (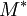 ) with their
) with their 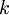 :
:
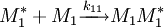
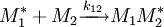
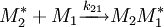
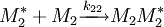
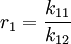
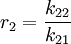
![\frac {d\left [M_1 \right]}{d\left [M_2\right]}=\frac{\left [M_1\right]\left (r_1\left[M_1\right]+\left [M_2\right]\right)}{\left [M_2\right]\left (\left [M_1\right]+r_2\left [M_2\right]\right)}](images/math/d/e/a/dea8c560dd0363f25dc92f414a5b64f6.png)



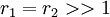 with both reactivity ratios very high the two monomers have no inclination to react to each other except with themselves leading to a mixture of two
with both reactivity ratios very high the two monomers have no inclination to react to each other except with themselves leading to a mixture of two 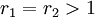 with both ratio's larger than 1, homopolymerization of component M_1 is favored but in the event of a crosspolymerization by M_2 the chain-end will continue as such giving rise to block copolymer
with both ratio's larger than 1, homopolymerization of component M_1 is favored but in the event of a crosspolymerization by M_2 the chain-end will continue as such giving rise to block copolymer
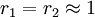 with both ratio's around 1, monomer 1 will react as fast with another monomer 1 or monomer 2 and a random copolymer results.
with both ratio's around 1, monomer 1 will react as fast with another monomer 1 or monomer 2 and a random copolymer results.
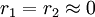 with both values approaching 0 the monomers are unable to react in homopolymerization and the result is an alternating polymer
with both values approaching 0 the monomers are unable to react in homopolymerization and the result is an alternating polymer
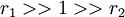 In the initial stage of the copolymerization monomer 1 is incorporated faster and the copolymer is rich in monomer 1. When this monomer gets depleted, more monomer 2 segments are added. This is called composition drift.
In the initial stage of the copolymerization monomer 1 is incorporated faster and the copolymer is rich in monomer 1. When this monomer gets depleted, more monomer 2 segments are added. This is called composition drift.
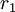 = 0.08) & cis-stilbene (
= 0.08) & cis-stilbene (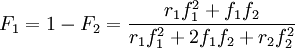
 stands the
stands the 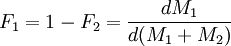
 the mole fraction of each monomer in the feed:
the mole fraction of each monomer in the feed:
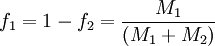
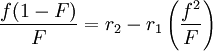
![f = \frac{[M_1]}{[M_2]} \,](images/math/5/7/a/57ac5143d99e53b5e8306a154f6cd38c.png) in the feed
in the feed
![F = \frac{d[M_1]}{d[M_2]} \,](images/math/b/b/c/bbc930eec4a70fde6beec25cd2ee4320.png) in the copolymer
in the copolymer
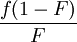 versus
versus 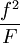 gives a straight line with slope
gives a straight line with slope 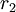 .
.
![\frac{-d[M_1]}{dt} = k_{11}[M_1]\sum[M_1^*] + k_{21}[M_1]\sum[M_2^*] \,](images/math/3/d/c/3dc78c19a18377553151beae43a910e4.png)
![\sum[M_x^*]](images/math/b/b/d/bbd242d963a5c6db2d1c721018379467.png) the concentration of all the active centers terminating in monomer 1 or 2.
the concentration of all the active centers terminating in monomer 1 or 2.
![\frac{-d[M_2]}{dt} = k_{22}[M_2]\sum[M_2^*] + k_{21}[M_2]\sum[M_1^*] \,](images/math/5/f/8/5f8cce20a1b811e90aa2818a1c6c01f3.png)
![\frac{d[M_1]}{d[M_2]} = \frac{[M_1]}{[M_2]} \left( \frac{k_{11}\frac{\sum[M_1^*]}{\sum[M_2^*]} + k_{21}} {k_{12}\frac{\sum[M_2^*]}{\sum[M_2^*]} + k_{22}} \right) \,](images/math/a/1/b/a1b85c579c466fc915181d71d62b3123.png)
![\frac{d\sum[M_1^*]}{dt} = \frac{d\sum[M_2^*]}{dt} \approx 0\,](images/math/a/0/d/a0d91130d633c58b23337bfd2ca3b8cb.png)
![k_{21}[M_1]\sum[M_2^*] = k_{12}[M_2]\sum[M_1^*] \,](images/math/1/6/6/166df310517c5eacaedeb43162a032f2.png)
![\frac{\sum[M_1^*]}{\sum[M_2^*]} = \frac{k_{21}[M_1]}{k_{12}[M_2]}\,](images/math/9/d/c/9dc9c686459d79d062be7dcd0a3d339b.png)


