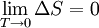To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Nernst heat theorem
The Nernst heat theorem was formulated by Walther Nernst early in the twentieth century and was used in the development of the third law of thermodynamics. Product highlight
The theoremThe Nernst heat theorem says that as absolute zero is approached, the entropy change ΔS for a chemical or physical transformation approaches 0. This can be expressed mathematically as follows:
Another way of looking at the theorem is to start with the definition of the Gibbs free energy (G), G = H - TS, where H stands for enthalpy. For a change from reactants to products at constant temperature and pressure the equation becomes ΔG = ΔH − TΔS. In the limit of T = 0 the equation reduces to just ΔG = ΔH, as illustrated in the figure shown here, which is supported by experimental data.[2] However, it is known from thermodynamics that the slope of the ΔG curve is -ΔS. Since the slope shown here reaches the horizontal limit of 0 as T → 0 then the implication is that ΔS → 0, which is the Nernst heat theorem. The historical significance of the Nernst heat theorem is that it was later used by Max Planck to give the third law of thermodynamics, which is that the entropy of all pure, perfectly crystalline homogeneous materials is 0 at absolute zero. See also
For further reading
References and notes
Categories: Chemistry | Thermochemistry | Thermodynamics |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Nernst_heat_theorem". A list of authors is available in Wikipedia. |