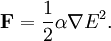To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Optical tweezersAn optical tweezer is a scientific instrument that uses a focused laser beam to provide an attractive or repulsive force, depending on the index mismatch (typically on the order of piconewtons) to physically hold and move microscopic dielectric objects. Optical tweezers have been particularly successful in studying a variety of biological systems in recent years. Product highlight
History and developmentThe detection of optical scattering and gradient forces on micrometer sized particles was first reported in 1970 by Arthur Ashkin[1], a scientist working at Bell Labs. Years later, Ashkin and colleagues reported the first observation[2] of what is now commonly referred to as an optical trap: a tightly focused beam of light capable of holding microscopic particles stable in three dimensions. One of the authors of this seminal 1986 paper, Steven Chu, would go on to use optical tweezing in his work on cooling and trapping atoms. This research earned Chu the 1997 Nobel Prize in Physics.[3]. In an interview[4], Steven Chu described how Askhin had first envisioned optical tweezing as a method for trapping atoms. Ashkin was able to trap larger particles (10 to 10,000 nanometers in diameter) but it fell to Chu to extend these techniques to the trapping of atoms (0.1 nanometers in diameter). In the late 1980s, Arthur Ashkin and his colleagues first applied the technology to the biological sciences, using it to trap an individual tobacco mosaic virus and Escherichia coli bacterium[5]. Throughout the 1990s and afterwards, researchers like Carlos Bustamante, James Spudich, and Steven Block pioneered the use of optical trap force spectroscopy to characterize molecular-scale biological motors. These molecular motors are ubiquitous in biology, and are responsible for locomotion and mechanical action within the cell. Optical traps allowed these biophysicists to observe the forces and dynamics of nanoscale motors at the single-molecule level; optical trap force-spectroscopy has since led to greater understanding of the stochastic nature of these force-generating molecules. Optical tweezers have proven useful in other areas of biology as well. For instance, in 2003 the techniques of optical tweezers were applied in the field of cell sorting; By creating a large optical intensity over the sample area filled with micro-biological sample, the cell can be sorted by its intrinsic optical characteristics.[6] [7]. In 2004 Optical tweezers made the leap from large, complicated, expensive machines to much simpler, smaller, inexpensive and ultimately portable systems with the introduction of DLBT (Diode Laser Bar Trapping) pioneered by Applegate et al. at the Colorado School of Mines[8]. Optical tweezers have also been used to probe the cytoskeleton, measure the visco-elastic properties of biopolymers, and study cell motility. The physics of optical tweezers
General descriptionOptical tweezers are capable of manipulating nanometer and micrometer-sized dielectric particles by exerting extremely small forces via a highly focused laser beam. The beam is typically focused by sending it through a microscope objective. The narrowest point of the focused beam, known as the beam waist, contains a very strong electric field gradient. It turns out that dielectric particles are attracted along the gradient to the region of strongest electric field, which is the center of the beam. The laser light also tends to apply a force on particles in the beam along the direction of beam propagation. It is easy to understand why if you imagine light to be a group of tiny particles, each impinging on the tiny dielectric particle in its path. This is known as the scattering force and results in the particle being displaced slightly downstream from the exact position of the beam waist, as seen in the figure. Optical traps are very sensitive instruments and are capable of the manipulation and detection of sub-nanometer displacements for sub-micrometre dielectric particles.[9] For this reason, they are often used to manipulate and study single molecules by interacting with a bead that has been attached to that molecule. DNA and the proteins and enzymes that interact with it are commonly studied in this way. For quantitative scientific measurements, most optical traps are operated in such a way that the dielectric particle rarely moves far from the trap center. The reason for this is that the force applied to the particle is linear with respect to its displacement from the center of the trap as long as the displacement is small. In this way, an optical trap can be compared to a simple spring, which follows Hooke's law. Detailed view of optical tweezersProper explanation of optical trapping behavior depends upon the size of the trapped particle relative to the wavelength of light used to trap it. In cases where the dimensions of the particle are greater than this wavelength, a simple ray optics treatment is sufficient. On the other hand, if the wavelength of light exceeds the particle dimensions, then the particles must be treated as tiny electric dipoles in an electric field. The ray optics approachIn cases where the diameter of a trapped particle is significantly greater than the wavelength of light, the trapping phenomenon can be explained using ray optics. As shown in the figure, individual rays of light emitted from the laser will be refracted as it enters and exits the dielectric bead. As a result, the ray will exit in a direction different from which it originated. Since light has a momentum associated with it, this change in direction indicates that its momentum has changed. Due to Newton's third law, there should be an equal and opposite momentum change on the particle. Most optical traps operate with a Gaussian beam (TEM00 mode) profile intensity. In this case, if the particle is displaced from the center of the beam, as in (a) in the figure, the particle has a net force returning it to the center of the trap because more intense beams impart a larger momentum change towards the center of the trap than less intense beams, which impart a smaller momentum change away from the trap center. The net momentum change, or force, returns the particle to the trap center. If the particle is located at the center of the beam, then individual rays of light are refracting through the particle symmetrically, resulting in no net lateral force. The net force in this case is along the axial direction of the trap, which cancels out the scattering force of the laser light. The cancellation of this axial gradient force with the scattering force is what causes the bead to be stably trapped slightly downstream of the beam waist. The electric dipole approximationIn cases where the diameter of a trapped particle is significantly smaller than the wavelength of light, the conditions for Rayleigh scattering are satisfied and the particle can be treated as a point dipole in an inhomogenous electromagnetic field. The force applied on a dipole in an electromagnetic field is known as the Lorentz force, The force on the dipole can be calculated by substituting two terms for the electric field in the equation above, one for each charge. The polarization of a dipole is Notice that the where in the second equality, it has been assumed that the dielectric particle is linear (i.e. In the final steps, two equalities will be used: (1) A Vector Analysis Equality, (2) One of Maxwell's Equations. First, the vector equality will be inserted for the first term in the force equation above. Maxwell's equation will be substituted in for the second term in the vector equality. Then the two terms which contain time derivatives can be combined into a single term.[10] The second term in the last equality is the time derivative of a quantity that is related through a multiplicative constant to the Poynting vector, which describes the power per unit area passing through a surface. Since the power of the laser is constant when sampling over frequencies much shorter than the frequency of the laser's light ~1013 Hz, the derivative of this term averages to zero and the force can be written as The square of the magnitude of the electric field is equal to the intensity of the beam as a function of position. Therefore, the result indicates that the force on the dielectric particle, when treated as a point dipole, is proportional to the gradient along the intensity of the beam. In other words, the gradient force described here tends to attract the particle to the region of highest intensity. In reality, the scattering force of the light works against the gradient force in the axial direction of the trap, resuling in an equilibrium position that is displaced slightly downstream of the intensity maximum. The scattering force depends linearly on the intensity of the beam, the cross section of the particle and the index of refraction of the trapping medium (e.g. water).[citation needed] Experimental design, construction and operationThe most basic optical tweezer setup will likely include the following components: a laser (usually Nd:YAG), a beam expander, some optics used to steer the beam location in the sample plane, a microscope objective and condenser to create the trap in the sample plane, a position detector (e.g. quadrant photodiode) to measure beam displacements and a microscope illumination source coupled to a CCD camera. The Nd:YAG laser (1064 nm wavelength) is the most common laser choice because biological specimens are most transparent to laser wavelengths around 1000 nm. This assures as low an absorption coefficient as possible, minimizing damage to the specimen, sometimes referred to as opticution. Perhaps the most important consideration in optical tweezer design is the choice of the objective. A stable trap requires that the gradient force, which depends upon the numerical aperture (NA) of the objective, be greater than the scattering force. Suitable objectives typically have a NA between 1.2 and 1.4.[11] While alternatives are available, perhaps the simplest method for position detection involves imaging the trapping laser exiting the sample chamber onto a quadrant photodiode. Lateral deflections of the beam are measured similarly to how its done using atomic force microscopy (AFM). Expanding the beam emitted from the laser to fill the aperture of the objective will result in a tighter, diffraction-limited spot.[12] While lateral translation of the trap relative to the sample can be accomplished by translation of the microscope slide, most tweezer setups have additional optics designed to translate the beam to give an extra degree of translational freedom. This can be done by translating the first of the two lenses labelled as "Beam Steering" in the figure. For example, translation of that lens in the lateral plane will result in a laterally deflected beam from what is drawn in the figure. If the distance between the beam steering lenses and the objective are chosen properly, this will correspond to a similar deflection before entering the objective and a resulting lateral translation in the sample plane. The position of the beam waist, that is the focus of the optical trap, can be adjusted by an axial displacement of the initial lens. Such an axial displacement causes the beam to diverge or converse slightly, the end result of which is an axially displaced position of the beam waist in the sample chamber.[13] Visualization of the sample plane is usually accomplished through illumination via a separate light source coupled into the optical path in the opposite direction using dichroic mirrors. This light is incident on a CCD camera and can be viewed on an external monitor or used for tracking the trapped particle position via video tracking. Descriptions of various optical tweezer setupsOptical tweezers based on alternate laser beam modesThe majority of optical tweezers make use of conventional TEM00 Gaussian beams. However a number of other beam types have been used to trap particles, including high order laser beams i.e Hermite Gaussian beam (TEMxy), Laguerre-Gaussian (LG) beams (TEMpl) and Bessel beams. Optical tweezers based on Laguerre Gaussian beam have the unique capability of trapping particles that are optically reflective and absorptive. Laguerre-Gaussian beams also possess a well-defined orbital angular momentum that can rotate particles.[14][15] This is accomplished without external mechanical or electrical steering of the beam. Both zeroth and higher Bessel Beams also possess a unique tweezing ability. They can trap and rotate multiple particles that are millimeters apart and even around obstacles. [16] Micromachines can be driven by these unique optical beams due to their intrinsic rotating mechanism due to the spin and orbital angular momentum of light.[citation needed] Multiplexed optical tweezersA typical setup uses one laser to create one or two traps. More complex optical tweezing operations can be achieved either by time-sharing a single laser beam among several optical tweezers or by diffractively splitting the beam into multiple traps. With acousto-optic deflectors or galvanometer-driven mirrors, a single laser beam can be shared among hundreds of optical tweezers in the focal plane, or else spread into an extended one-dimensional trap. Specially designed diffractive optical elements can divide a single input beam into hundreds of continuously illuminated traps in arbitrary three-dimensional configurations. The trap-forming hologram also can specify the mode structure of each trap individually, thereby creating arrays of optical vortices, optical tweezers, and holographic line traps, for example. When implemented with a spatial light modulator, such holographic optical traps also can move objects in three dimensions. Optical tweezers based on optical fibersThe fiber optical trap relies on the same principle as the optical trapping, but with the laser delivered through an Optical fiber. If one end of the optical fiber tip is moulded into a lens-like facet, that lens tip will act as a focusing (converging) point for the high optical gradient trap to be formed.[17] On the other hand, if the ends of the fiber are not moulded, the laser exiting the fiber will be diverging and thus a stable optical trap can only be realised by balancing the gradient and the scattering force from two opposing ends of the fiber. The gradient force will trap the particles the transverse direction, while the axial optical force comes from the scattering force of the two counter propagating beams emerging from the two fibers. The equilibrium z-position of such a trapped bead is where the two scattering forces equal each other. This work was pioneered by A. Constable et al., Opt. Lett. 18,1867 (1993), and followed by J.Guck et al., Phys. Rev. Lett. 84, 5451 (2000), who made use of this technique to stretch microparticles. By manipulating the input power into the two ends of the fiber, there will be an increase of a "optical stretching" that can be used to measure viscoelastic properties of cells, with sensitivity sufficient to distinguish between different individual cytoskeletal phenotypes. i.e. human erythrocytes and mouse fibroblasts. A recent test has seen great success in differentiating cancerous cells from non-cancerous ones from the two opposed, non-focused laser beams. [18] Optical tweezers in a 'landscape' (cell sorting)One of the more common cell-sorting systems makes use of flow cytometry through fluorescent imaging. In this method, a suspension of biologic cells is sorted into two or more containers, based upon specific fluorescent characteristics of each cell during an assisted flow. By using an electrical charge that the cell is "trapped" in, the cells are then sorted based on the fluorescence intensity measurements. The sorting process is undertaken by an electrostatic deflection system that diverts cells into containers based upon their charge. In the optically-actuated sorting process, the cells are flowed through into an optical landscape i.e. 2D or 3D optical lattices. Without any induced electrical charge, the cells would sort based on their intrinsic refractive index properties and can be re-configurability for dynamic sorting. Mike MacDonald, Gabe Spalding and Kishan Dholakia, Nature 426, 421-424 (2003)[1] made use of diffractive optics and optical elements to create the optical lattice. An automated cell sorter was described at the University of Toronto in 2001, but made use of mechanical parameters as opposed to spatial light modulation [19] On the other hand, K. Ladavac, K. Kasza and D. G. Grier, Physical Review E 70, 010901(R) (2004)[2] made use of the spatial light modulator to project an intensity pattern to enable the optical sorting process. The main mechanism for sorting is the arrangement of the optical lattice points. As the cell flow through the optical lattice, there are forces due to the particles drag force that is competing directly with the optical gradient force (See Physics of an Optical Tweezers) from the optical lattice point. By shifting the arrangement of the optical lattice point, there is a preferred optical path where the optical forces are dominant and biased. With the aid of the flow of the cells, there is a resultant force that is directed along that preferred optical path. Hence, there is a relationship of the flow rate with the optical gradient force. By adjusting the two forces, one will be able to obtain a good optical sorting efficiency. Competition of the forces in the sorting environment need fine tuning to succeed in high efficient optical sorting. The need is mainly with regards to the balance of the forces; drag force due to fluid flow and optical gradient force due to arrangement of intensity spot. Scientists at the University of St. Andrews have received considerable funding from the UK Engineering and Physical Sciences Research Council (EPSRC) for an optical sorting machine. This new technology could rival the conventional fluorescence-activated cell sorting.[20] Optical tweezers based on evanescent fieldsAn evanescent field [3] [4] is a residue optical field that "leaks" during total internal reflection. This "leaking" of light fades off at an exponential rate. The evanescent field has found a number of applications in nanometer resolution imaging (microscopy); optical micromanipulation (optical tweezers) are becoming ever more relevant in research. In optical tweezers, a continuous evanescent field can be created when light is propagating through an optical waveguide (multiple total internal reflection). The resulting evanescent field has a directional sense and will propel microparticles along its propagating path. This work was first pioneered by S. Kawata and T. Sugiura, in 1992 (Opt. Lett. 17 (11), 772 (1992)). Kawata showed that the field can be coupled to the particles in proximity on the order of 100 nanometers. This direct coupling of the field is treated as a type of photon tunnelling across the gap from prism to microparticles. The result is a directional optical propelling force. A recent updated version of the evanescent field optical tweezers make use of extended optical landscape patterns to simultaneously guide a large number of particles into a preferred direction without using a waveguide. It is termed as Lensless Optical Trapping (“LOT”) [5]. The orderly movement of the particles is aided by the introduction of Ronchi Ruling that creates well-defined optical potential wells (replacing the waveguide). This means that particles are propelled by the evanescent field while being trapped by the linear bright fringes. At the moment, there are scientists working on focused evanescent fields as well. Optical tweezers: an indirect approachMing Wu, a UC Berkeley Professor of electrical engineering and computer sciences invented the new optoelectronic tweezers. Wu transformed the optical energy from low powered light emitting diodes (LED) into electrical energy via a photoconductive surface. The ideas is to allow the LED to switch on and off the photoconductive material via its fine projection. As the optical pattern can be easily transformable through optical projection, this method allow a high flexibility of switching different optical landscapes. The manipulation/tweezing process is done by the variations between the electric field actuated by the light pattern. As the particles will be either attracted or repelled from the actuated point due to the its induced electrical dipole. Particles being suspended in a liquid will be susceptible to electrical field gradient, this is known as dielectrophoresis. One clear advantage is that the electrical conductivity between a different cells. Living cells have a lower conductive medium while the dead ones have minimum or no conductive medium. The system may be able to manipulate roughly 10,000 cells or particles at the same time. See comments by Professor Kishan Dholakia on this new technique, K. Dholakia, Nature Materials 4, 579-580 (01 Aug 2005) News and Views. "The system was able to move live E. coli bacteria and 20-micrometre-wide particles, using an optical power output of less than 10 microwatts. This is one-hundred-thousandth of the power needed for [direct] optical tweezers"[6]. Optical bindingWhen a cluster of microparticles are trapped within a monochromatic laser beam, the organization of the microparticles within the optical trapping is heavily dependent on the redistributing of the optical trapping forces amongst the microparticles. This redistribution of light forces amongst the cluster microparticles provides a new force equilibrium on the cluster as a whole. As such we can say that the cluster of microparticles are somewhat bounded together by light. One of the first evidence of optical binding was reported by Michael M. Burns, Jean-Marc Fournier, and Jene A. Golovchenko [7]. Research goups using optical tweezersHere is a list of research groups using optical tweezers and the type of research they perform.
Other resourcesExtensive link pages to other academic groups in optical tweezing
Professional papers reviewing optical tweezers
Web resources
Multimedia links
Commercial optical tweezer systemsCommercial suppliers of optical tweezer systems include (in alphabetical order)
References
Categories: Photonics | Condensed matter physics |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Optical_tweezers". A list of authors is available in Wikipedia. |





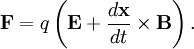
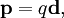 where
where 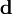 is the distance between the two charges. For a point dipole, the distance is infinitesimal,
is the distance between the two charges. For a point dipole, the distance is infinitesimal, 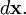 Taking into account that the two charges have opposite signs, the force takes the form
Taking into account that the two charges have opposite signs, the force takes the form
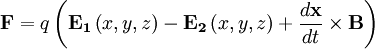
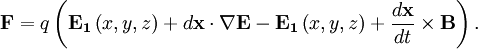
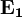 cancel out. Multiplying through by the charge,
cancel out. Multiplying through by the charge, 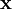 , into polarization,
, into polarization, 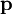 ,
,
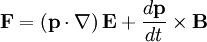
![\mathbf{F}=\alpha\left[\left(\mathbf{E}\cdot\nabla\right)\mathbf{E}+\frac{d\mathbf{E}}{dt}\times\mathbf{B}\right],](images/math/4/f/3/4f396ec71310bb2ae4643f9ae243059f.png)
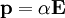 ).
).
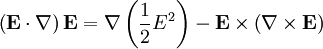
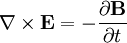
![\mathbf{F}=\alpha\left[\frac{1}{2}\nabla E^2-\mathbf{E}\times\left(\nabla\times\mathbf{E}\right)+\frac{d\mathbf{E}}{dt}\times\mathbf{B}\right]](images/math/9/a/1/9a184c3c8d20f3aee881326d0dd3c581.png)
![\mathbf{F}=\alpha\left[\frac{1}{2}\nabla E^2-\mathbf{E}\times\left(-\frac{d\mathbf{B}}{dt}\right)+\frac{d\mathbf{E}}{dt}\times\mathbf{B}\right]](images/math/b/c/4/bc4cf96eca6e5d713d40c9e595fdb142.png)
![\mathbf{F}=\alpha\left[\frac{1}{2}\nabla E^2+\frac{d}{dt}\left(\mathbf{E}\times\mathbf{B}\right)\right].](images/math/9/3/2/9326d17f5491ac310f3cc5811c85ee3e.png)