To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Redox electrodeA redox electrode is an electrode made from electron-conductive material and characterized by high chemical stability in the solution under test. It is used for measuring the redox potential of a specific redox system in solution, and for electrochemical studies (investigation of electrochemical kinetics of interfacial processes). Product highlightThe correlation of an electrode potential and redox system composition can be described by the Nernst equation:
where
There are two general classes of redox electrodes:
IllustrationA gold disk redox electrode with a Teflon shroud insulating the disk. See alsoCategories: Electrodes | Electrochemistry |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Redox_electrode". A list of authors is available in Wikipedia. |





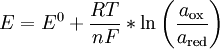 ,
,


