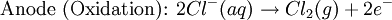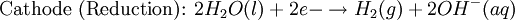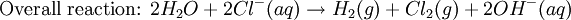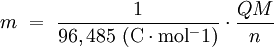To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Electrochemistry
Electrochemistry is a branch of chemistry that studies chemical reactions which take place at the interface of an electron conductor (the electrode composed of a metal or a semiconductor, including graphite) and an ionic conductor (the electrolyte) and which involve electron transfer between the electrode and the electrolyte. If a chemical reaction is caused by an external voltage, as in electrolysis, or if a voltage is caused by a chemical reaction, as in a battery, it is an electrochemical reaction. Chemical reactions where electrons are transferred between atoms are called oxidation/reduction (redox) reactions. In general, electrochemistry deals with situations where oxidation and reduction reactions are separated in space, connected by an external electric circuit. Product highlight
History16th to 18th century developmentsThe 16th century marked the beginning of electrical understanding. During that century the English scientist William Gilbert spent 17 years experimenting with magnetism and, to a lesser extent, electricity. For his work on magnets, Gilbert became known as the "Father of Magnetism." He discovered various methods for producing and strengthening magnets. In 1663 the German physicist Otto von Guericke created the first electric generator, which produced static electricity by applying friction in the machine. The generator was made of a large sulfur ball cast inside a glass globe, mounted on a shaft. The ball was rotated by means of a crank and a static electric spark was produced when a pad was rubbed against the ball as it rotated. The globe could be removed and used as source for experiments with electricity. By the mid—1700s the French chemist Charles François de Cisternay du Fay discovered two types of static electricity, and that like charges repel each other whilst unlike charges attract. Du Fay announced that electricity consisted of two fluids: "vitreous" (from the Latin for "glass"), or positive, electricity; and "resinous," or negative, electricity. This was the two-fluid theory of electricity, which was to be opposed by Benjamin Franklin's one-fluid theory later in the century. Charles-Augustin de Coulomb developed the law of electrostatic attraction in 1781 as an outgrowth of his attempt to investigate the law of electrical repulsions as stated by Joseph Priestley in England. In the late 1700s the Italian physician and anatomist Luigi Galvani marked the birth of electrochemistry by establishing a bridge between chemical reactions and electricity on his essay "De Viribus Electricitatis in Motu Musculari Commentarius" (Latin for Commentary on the Effect of Electricity on Muscular Motion) in 1791 where he proposed a "nerveo-electrical substance" on biological life forms. On his essay Galvani concluded that animal tissue contained a here-to-fore neglected innate, vital force, which he termed "animal electricity," which activated nerves and muscles spanned by metal probes. He believed that this new force was a form of electricity in addition to the "natural" form produced by lightning or by the electric eel and torpedo ray as well as the "artificial" form produced by friction (i.e., static electricity). Galvani's scientific colleagues generally accepted his views, but Alessandro Volta rejected the idea of an "animal electric fluid," replying that the frog's legs responded to differences in metal temper, composition, and bulk. Galvani refuted this by obtaining muscular action with two pieces of the same material. 19th centuryIn 1800, the English chemists William Nicholson and Johann Wilhelm Ritter succeeded in decomposing water into hydrogen and oxygen by electrolysis. Soon thereafter Ritter discovered the process of electroplating. He also observed that the amount of metal deposited and the amount of oxygen produced during an electrolytic process depended on the distance between the electrodes. By 1801 Ritter observed thermoelectric currents and anticipated the discovery of thermoelectricity by Thomas Johann Seebeck. By the 1810s William Hyde Wollaston made improvements to the galvanic pile. Sir Humphry Davy's work with electrolysis led to the conclusion that the production of electricity in simple electrolytic cells resulted from chemical action and that chemical combination occurred between substances of opposite charge. This work led directly to the isolation of sodium and potassium from their compounds and of the alkaline earth metals from theirs in 1808. Hans Christian Ørsted's discovery of the magnetic effect of electrical currents in 1820 was immediately recognized as an epoch-making advance, although he left further work on electromagnetism to others. André-Marie Ampère quickly repeated Ørsted's experiment, and formulated them mathematically. In 1821, Estonian-German physicist Thomas Johann Seebeck demonstrated the electrical potential in the juncture points of two dissimilar metals when there is a heat difference between the joints. In 1827 the German scientist Georg Ohm expressed his law in this famous book "Die galvanische Kette, mathematisch bearbeitet" (The Galvanic Circuit Investigated Mathematically) in which he gave his complete theory of electricity. In 1832 Michael Faraday's experiments led him to state his two laws of electrochemistry. In 1836 John Daniell invented a primary cell in which hydrogen was eliminated in the generation of the electricity. Daniell had solved the problem of polarization. In his laboratory he had learned that alloying the amalgamated zinc of Sturgeon with mercury would produce a better voltage. William Grove produced the first fuel cell in 1839. In 1846, Wilhelm Weber developed the electrodynamometer. In 1866, Georges Leclanché patented a new cell which eventually became the forerunner to the world's first widely used battery, the zinc carbon cell. Svante August Arrhenius published his thesis in 1884 on Recherches sur la conductibilité galvanique des électrolytes (Investigations on the galvanic conductivity of electrolytes). From his results the author concluded that electrolytes, when dissolved in water, become to varying degrees split or dissociated into electrically opposite positive and negative ions. In 1886 Paul Héroult and Charles M. Hall developed a successful method to obtain aluminum by using the principles described by Michael Faraday. In 1894 Friedrich Ostwald concluded important studies of the electrical conductivity and electrolytic dissociation of organic acids. Walther Hermann Nernst developed the theory of the electromotive force of the voltaic cell in 1888. In 1889, he showed how the characteristics of the current produced could be used to calculate the free energy change in the chemical reaction producing the current. He constructed an equation, known as Nernst Equation, which related the voltage of a cell to its properties. In 1898 Fritz Haber showed that definite reduction products can result from electrolytic processes if the potential at the cathode is kept constant. In 1898 he explained the reduction of nitrobenzene in stages at the cathode and this became the model for other similar reduction processes. The 20th century and recent developmentsIn 1902, The Electrochemical Society (ECS) was founded. In 1909, Robert Andrews Millikan began a series of experiments to determine the electric charge carried by a single electron. In 1923, Johannes Nicolaus Brønsted and Thomas Martin Lowry published essentially the same theory about how acids and bases behave, using an electrochemical basis. Arne Tiselius developed the first sophisticated electrophoretic apparatus in 1937 and some years later he was awarded the 1948 Nobel Prize for his work in protein electrophoresis. A year later, in 1949, the International Society of Electrochemistry (ISE) was founded. By the 1960s–1970s quantum electrochemistry was developed by Revaz Dogonadze and his pupils. PrinciplesRedox reactionsElectrochemical processes involve redox reactions where energy is produced by a spontaneous reaction which produces electricity, or where electrical current stimulates a chemical reaction. In a redox reaction, an atom's or ion's oxidation state (basically, its charge) changes as a result of an electron transfer. Oxidation and ReductionThe elements involved in an electrochemical reaction are characterized by the number of electrons each has. The oxidation state of an ion is the number of electrons it has accepted or donated compared to its neutral state (which is defined as having an oxidation state of 0). If an atom or ion donates an electron in a reaction its oxidation state is increased. In the other direction, if an element accepts an electron its oxidation state is decreased. In general, oxidation and reduction always occur in a paired fashion such that one species is oxidized when another is reduced. A reaction in which both oxidation and reduction is occurring is called a redox reaction. For example when sodium reacts with chlorine, sodium donates one electron and attains an oxidation state of +1. Chlorine accepts the electron and attains an oxidation state of −1. The sign of the oxidation state (positive/negative) actually corresponds to the value of each ion's electronic charge. The attraction of the differently charged sodium and chlorine ions is the reason they then form an ionic bond. The loss of electrons from an atom or molecule is called oxidation, and the gain of electrons is reduction. This can be easily remembered through the use of mnemonic devices. Two of the most popular are "OIL RIG" (Oxidation Is Loss, Reduction Is Gain) and "LEO" the lion says "GER" (Lose Electrons: Oxidization, Gain Electrons: Reduction). For cases where electrons are shared (covalent bonds) between atoms with large differences in electronegativity, the electron is assigned to the atom with the largest electronegativity in determining the oxidation state. The atom or molecule which loses electrons is known as the reducing agent, or reductant, and the substance which accepts the electrons is called the oxidizing agent, or oxidant. The oxidizing agent is always being reduced in a reaction; the reducing agent is always being oxidized. Oxygen is an oxidant, but not the only one. Despite the name, an oxidation reaction does not necessarily need to involve oxygen. In fact, even fire can be fed by an oxidant other than oxygen: fluorine fires are often unquenchable, as fluorine is an even stronger oxidant (it has a higher electronegativity) than oxygen. For reactions involving oxygen, the gain of oxygen implies the oxidation of the atom or molecule to which the oxygen is added (the oxygen is reduced). This follows since oxygen is highly electronegative (this can be extended to include other highly electronegative atoms such a fluorine). In compounds involving non-metals, the loss of hydrogen implies oxidation of the atom or molecule from which it is lost (hydrogen is reduced). This follows because the hydrogen donates its electron in covalent bonds with non-metals but it takes the electron along when it is lost. Conversely, loss of oxygen or gain of hydrogen implies reduction. Electrochemical cellsAn electrochemical cell is a device capable of producing electric current from energy released by a spontaneous redox reaction. This kind of cell includes the Galvanic cell or Voltaic cell, named after Luigi Galvani and Alessandro Volta, both scientists who conducted several experiments on chemical reactions and electric current during the late 18th century. Electrochemical cells have two electrodes (the anode and the cathode). The anode is defined as the electrode where oxidation occurs and the cathode is the electrode where the reduction takes place. Electrodes come in various forms including metal, gas and carbon. The Galvanic cell uses two different metal electrodes, each in an electrolyte where the cations are the oxidized form of the electrode metal. The tendency of the electrode metals to oxidize or reduce, in a particular electrolyte, is defined in terms of the electrochemical potential which depends on the temperature, pressure, and the componsition and concentration of the electrolyte. In a particular cell, one electrode will undergo oxidation (the anode) and the other will undergo reduction (the cathode), depending on the sum of the electrochemical potential differences at both electrodes. Thus, a cell will produce an electrical potential difference between the two electrodes whose magnitude and sign depends on the chemistry occurring at both electrodes. A Galvanic cell whose electrodes are zinc and copper submerged in zinc sulfate and copper sulfate, respectively, is known as a Daniells cell. Half reactions for a Daniells cell are these:
To provide a complete electric circuit, there must be a conduction path between the anode and cathode electrolytes. The simplest ionic conduction path is to provide a liquid junction. To avoid convective mixing between the two electrolytes, the liquid junction can be provided through a porous plug that allows ion flow while restricting convective flow. Such porous plug connections allow the anions and cations from the two electrolytes to mix, which can cause contamination. To minimize such issues, a salt bridge can be used which consists of an electrolyte saturated gel in an inverted U-tube. Contamination issues can be minimized by careful choices of the electrolytes in the anode, cathode and salt bridge. A voltmeter is capable of measuring the change of electrical potential between the anode and the cathode. Electrochemical cell voltage is also referred to as electromotive force or emf. A cell diagram can be used to trace the path of the electrons in the electrochemical cell. For example, here is a cell diagram of a Daniells cell: First, the reduced form of the metal to be oxidized at the anode (Zn) is written. This is separated from its oxidized form by a vertical line, which represents the limit between the phases (oxidation changes). The double vertical lines represent the saline bridge on the cell. Finally, the oxidized form of the metal to be reduced at the cathode, is written, separated from its reduced form by the vertical line. The electrolyte concentration is given as it is an important variable in determining the cell potential. Standard electrode potentialTo allow prediction of the cell potential, tabulations of standard electrode potential are available. Such tabulations are referenced to the standard hydrogen electrode (SHE). The standard hydrogen electrode undergoes the reaction which is shown as reduction but, in fact, the SHE can act as either the anode or the cathode, depending on the relative oxidation/reduction potential of the other electrode/electrolyte combination. The term standard in SHE requires a supply of hydrogen gas bubbled through the electrolyte at a pressure of 1 atm and an acidic electrolyte with H+ activity equal to 1 (usually assumed to be [H+] = 1 mol/liter). The SHE electrode can be connected to any other electrode by a salt bridge to form a cell. If the second electrode is also at standard conditions, then the measured cell potential is called the standard electrode potential for the electrode. The standard electrode potential for the SHE is zero, by definition. The polarity of the standard electrode potential provides information about the relative reduction potential of the electrode compared to the SHE. If the electrode has a positive potential with respect to the SHE, then that means it is a strongly reducing electrode which forces the SHE to be the anode (an example is Cu in aqueous CuSO4 with a standard electrode potential of 0.337 V). Conversely, if the measured potential is negative, the electrode is more oxidizing than the SHE (such as Zn in ZnSO4 where the standard electrode potential is -0.763 V). Standard electrode potentials are usually tabulated as reduction potentials. However, the reactions are reversible and the role of a particular electrode in a cell depends on the relative oxidation/reduction potential of both electrodes. The oxidation potential for a particular electrode is just the negative of the reduction potential. A standard cell potential can be determined by looking up the standard electrode potentials for both electrodes (sometimes called half cell potentials). The one that is smaller will be the anode and will undergo oxidation. The cell potential is then calculated as the sum of the reduction potential for the cathode and the oxidation potential for the anode. For example, the standard electrode potential for a copper electrode is: At standard temperature, pressure and concentration conditions, the cell's emf (measured by a multimeter) is 0.34 V. by definition, the electrode potential for the SHE is zero. Thus, the Cu is the cathode and the SHE is the anode giving Or, Changes in the stoichiometric coefficients of a balanced cell equation will not change Spontaneity of Redox reactionDuring operation of electrochemical cells, chemical energy is transformed into electrical energy and is expressed mathematically as the product of the cell's emf and the electrical charge transferred through the external circuit. where The emf of the cell at zero current is the maximum possible emf. It is used to calculate the maximum possible electrical energy that could be obtained from a chemical reaction. This energy is referred as electrical work and is expressed by the following equation: where work is defined as positive into the system. Since the free energy is the maximum amount of work that can be extracted from a system, one can write: A positive cell potential gives a negative change in Gibbs free energy. This is consistent with the cell production of an electric current flowing from the cathode to the anode through the external circuit. If the current is driven in the opposite direction by imposing an external potential, then work is done on the cell to drive electrolysis. A spontaneous electrochemical reaction (change in Gibbs free energy less than zero) can be used to generate an electric current, in electrochemical cells. This is the basis of all batteries and fuel cells. For example, gaseous oxygen (O2) and hydrogen (H2) can be combined in a fuel cell to form water and energy, typically a combination of heat and electrical energy. Conversely, non-spontaneous electrochemical reactions can be driven forward by the application of a current at sufficient voltage. The electrolysis of water into gaseous oxygen and hydrogen is a typical example. The relation between the equilibrium constant and the Gibbs free energy for an electrochemical cell is expressed as follows: Rearranging to express the relation between standard potential and equilibrium constant yields Previous equation can use Briggsian logarithm as shown below: Cell emf dependency on changes in concentrationNernst EquationThe standard potential of an electrochemical cell requires standard conditions for all of the reactants. When reactant concentrations differ from standard conditions, the cell potential will deviate from the standard potential. In 1900s German chemist Walther Hermann Nernst proposed a mathematical model to determine the effect of reactant concentration on electrochemical cell potential. In the late 1800s Willard Gibbs formulated a theory to predict whether a chemical reaction is spontaneous based on the free energy
Where: ΔG = change in Gibbs free energy, T = absolute temperature, R = gas constant, ln = natural logarithm, Q = reaction quotient. Gibbs key contribution was to formalize the understanding of the effect of reactant concentration on spontaneity. Based on Gibbs's work, Nernst extended the theory to include the contribution from electric potential on charged species. As shown in the previous section, the change in Gibbs free energy for an electrochemical cell can be related to the cell potential. Thus, Gibbs theory becomes Where: n = number of electrons/mole product, F = Faraday constant (coulombs/mole), and ΔE = cell potential. Finally, Nernst divided through by the amount of charge transferred to arrive at a new equation which now bears his name: Assuming standard conditions ( Concentration cellsA concentration cell is an electrochemical cell where the two electrodes are the same material, the electrolytes on the two half-cells involve the same ions, but the electrolyte concentration differs between the two half-cells. For example an electrochemical cell, where two copper electrodes are submerged in blue vitriol's solution, whose concentrations are 0.05 M and 2.0 M, connected through a salt bridge. This type of cell will generate a potential that can be predicted by the Nernst equation. Both electrodes undergo the same basic chemistry (although the reaction proceeds in reverse at the cathode) Le Chatelier's principle indicates that the reaction is more favourable to reduction as the concentration of The following cell diagram describes the cell mentioned above: Where the half cell reactions for oxidation and reduction are: Where the cell's emf is calculated through Nernst equation as follows:
However, this value is only approximate, as reaction quotient is defined in terms of ion activities which can be approximated with the concentrations as calculated here. The Nernst equation plays an important role in understanding electrical effects in cells and organelles. Such effects include nerve synapses and cardiac beat as well as the resting potential of a somatic cell. BatteryA battery is an electrochemical cell (sometimes several in series) used for chemical energy storage. Batteries are optimized to produce a constant electric current for as long as possible. Although the cells discussed previously, including a salt bridge, are useful for theoretical purposes and some laboratory purposes, the large internal resistance of the salt bridge make them inappropriate battery technologies. Various alternative battery technologies have been commercialized as discussed next. Dry cellDry cells do not have a fluid electrolyte. Instead, they use a moist electrolyte paste. Leclanché's cell is a good example of this, where the anode is a zinc container surrounded by a thin layer of manganese dioxide and a moist electrolyte paste of ammonium chloride and zinc chloride mixed with starch. The cell's cathode is represented by a carbon bar inserted on the cell's electrolyte, usually placed in the middle. Leclanché's simplified half reactions are shown below: The voltage obtained from the zinc-carbon battery is around 1.5 V. Mercury batteryThe mercury battery has many applications in medicine and electronics. The battery consists of a steel—made container in the shape of a cylinder acting as the cathode, where an amalgamated anode of mercury and zinc is surrounded by a stronger alkaline electrolyte and a paste of Zinc oxide and Mercury(II) oxide . Mercury battery half reactions are shown below: There are no changes in the electrolyte's composition when the cell works. Such Mercurium batteries provide 1.35 V of direct current. Lead-acid battery
The Lead-acid battery used in automobiles, consists of a series of six identical cells assembled in series. Each cell has a lead anode and a cathode made from lead dioxide packed in a metal plaque. Cathode and anode are submerged in a solution of sulfuric acid acting as the electrolyte. Lead-acid battery half cell reactions are shown below:
At standard conditions, each cell may produce a potential of 2 V, hence overall voltage produced is 12 V. Differing from Mercury and Zinc-carbon batteries, lead-acid batteries are rechargeable. If an external voltage is supplied to the battery it will produce an electrolysis of the products in the overall reaction (discharge), thus recovering initial components which made the battery work. Solid state Lithium batteryInstead of an aqueous electrolyte or a moist electrolyte paste, a solid state battery operates using a solid electrolyte. Solid state lithium batteries are an example of this; a solid Lithium bar acts as the anode, a bar of Lithium sulfide or Vanadium oxide acts as the cathode, and a polymer, allowing the passage of ions and not electrons, serves as the electrolyte. The advantage of this kind of battery is that Lithium possess the highest negative value of standard reduction potential. It is also a light metal and therefore less mass is required to generate 1 mole of electrons. Lithium ion battery technologies are widely used in portable electronic devices because they have high energy storage density and are rechargeable. These technologies show promise for future automotive applications. Flow battery/ Redox flow batteryMost batteries have all of the electrolyte and electrodes within a single housing. A flow battery is unusual in that the majority of the electrolyte, including dissolved reactive species, is stored in separate tanks. The electrolytes are pumped through a reactor, which houses the electrodes, when the battery is charged or discharged. These types of batteries are typically used for large-scale energy storage (kWh - multi MWh). Of the several different types that have been developed, some are of current commercial interest, including the vanadium redox battery and zinc bromine battery. Fuel cellsFossil fuels are used in power plants to supply electrical needs, however their conversion into electricity is an inefficient process. The most efficient electrical power plant may only convert about 40% of the original chemical energy into electricity when burned or processed. To enhance electrical production, scientists have developed fuel cells where combustion is replaced by electrochemical methods, similar to a battery but requiring continuous replenishment of the reactants consumed. The most popular is the oxygen-hydrogen fuel cell, where two inert–electrodes (porous electrodes of Nickel and Nickel oxide) are placed in an electrolytic solution such as hot caustic potash, in both compartments (anode and cathode) gaseous hydrogen and oxygen are bubbled into solution. Oxygen-hydrogen fuel cell reactions are shown bellow: The overall reaction is identical to hydrogen combustion. Oxidation and reduction take place in the anode and cathode separately. This is similar to the electrode used in the cell for measuring standard reduction potential which has a double function acting as electrical conductors providing a surface required to decomposition of the molecules into atoms before electron transferring, thus named electrocatalysts. Platinum, nickel, rhodium are good electrocatalysts. CorrosionCorrosion is the term applied to metal rust caused by an electrochemical process. Most people are likely familiar with the corrosion of iron, in the form of reddish rust. Other examples include the black tarnish on silver, and red or green corrosion that may appear on copper and its alloys, such as brass. The cost of replacing metals lost to corrosion is in the multi-billions of dollars per year. Iron corrosionFor iron rust to occur the metal has to be in contact with oxygen and water, although chemical reactions for this process are relatively complex and not all of them are completely understood, it is believed the causes are the following:
Iron corrosion takes place on acid medium; H+ ions come from reaction between carbon dioxide in the atmosphere and water, forming carbonic acid. Fe2+ ions oxides, following this equation: Iron(III) oxide hydrated is known as rust. Water associated with iron oxide it varies, thus chemical representation is presented as Corrosion of coinage metalsCoinage metals, such as copper and silver, can also slowly corrode. At standard temperature and pressure, a patina of green-blue copper carbonate forms on the surface of copper. Silver cutlery that is in contact with food can develop a layer of Silver sulfide. Prevention of CorrosionAttempts to save a metal from becoming anodic are of two general types. Anodic regions dissolve and destroy the structural integrity of the metal. While it is almost impossible to prevent anode/cathode formation, if a non-conducting material covers the metal, contact with the electrolyte is not possible and corrosion will not occur. CoatingMetals are coated on its surface with paint or some other non-conducting coating. This prevents the electrolyte from reaching the metal surface if the coating is complete. Scratches exposing the metal will corrode with the region under the paint, adjacent to the scratch, to be anodic. Other prevention is called passivation where a metal is coated with another metal such as a tin can. Tin is a metal that rapidly corrodes to form a mono-molecular oxide coating that prevents further corrosion of the tin. The tin prevents the electrolyte from reaching the base metal, usually steel (iron). However, if the tin coating is scratched the iron becomes anodic and the can corrodes rapidly. Sacrificial anodesA method commonly used to protect a structural metal is to attach a metal which is more anodic than the metal to be protected. This forces the structural metal to be cathodic, thus spared corrosion. It is called "sacrificial" because the anode dissolves and has to be replaced periodically. Zinc bars are attached at various locations on steel ship hulls to render the ship hull cathodic. The zinc bars are replaced periodically. Other metals, such as magnesium, would work very well but zinc is the least expensive useful metal. To protect pipelines, buried or exposed an ingot of magnesium (or zinc) is buried beside the pipeline and connected electrically to the pipe above ground. The pipeline is forced to be a cathode and is protected. The magnesium anode is sacrificed. At intervals new ingots are buried to replace those lost. ElectrolysisSpontaneous redox reactions produces electricity, thus passage of electrons through a wire in the electric circuit. Electrolysis requires an external source of electrical energy to induce a chemical reaction, this process takes place in a compartment called electrolytic cell. Principles involved on electrolysis are the same as featured on electrochemical cells. Electrolysis of molten sodium chlorideWhen molten, sodium chloride can be electrolysed to yield metallic sodium and gaseous chlorine. Industrially this process takes place in a special cell named Down's cell. The cell is connected to a battery, allowing electrons migration from the battery to the electrolytic cell. Reactions that take place at Down's cell are the following: This process can yield industrial amounts of metallic sodium and gaseous chlorine, and is widely used on mineral dressing and metallurgy industries. Standard emf for this process is approximately -4 V indicating a non-spontaneous process. In order this reaction to occur the battery should provide at least a potential of 4V. However, on mineral refining industry, higher voltages are used, due to low efficiency of the process. Electrolysis of water
Water at standard temperature and pressure conditions doesn't decompose into hydrogen and oxygen spontaneously as the Gibbs free energy for the process at standard conditions is about 474.4 kJ However, special laboratory glassware has been designed for this purpose- the Hofmann voltameter. In it, a pair of inert electrodes usually made of platinum act as anode and cathode in the electrolytic process. After the water (if pure) has been placed in the apparatus, nothing happens, hence there are not enough ions to let the passage of electrons occur. To start the electrolysis an electrolyte should be placed in, usually sodium chloride or sulfuric acid (most used 0.1 M). Bubbles from the gases will be seen near both electrodes. The following half reactions describe the process mentioned above: Although strong acids may be used in the apparatus, the reaction will not net consume the acid. Electrolysis of aqueous solutionsElectrolysis in an aqueous is a similar process as mentioned in electrolysis of water. However, it is considered to be a complex process because the contents in solution have to be analyzed in half reactions, whether reduced or oxidized. Electrolysis of a solution of Sodium chlorideThe presence of water in a solution of sodium chloride must be examined in respect to its reduction and oxidation in both electrodes. Usually, water is electrolysed as mentioned in electrolysis of water yielding gaseous oxygen in the anode and gaseous hydrogen in the cathode. On the other hand, sodium chloride in water dissociates in Na+ and Cl- ions, anion will be attracted to the cathode, thus reducing the sodium ion. The cation will then be attracted to the anode oxidizing chloride ion. The following half reactions describes the process mentioned: Reaction 1 is discarded as it has the most negative value on standard reduction potential thus making it less thermodynamically favorable in the process. When comparing the reduction potentials in reactions 2 & 4, the reduction of chloride ion is favored. Thus, if the Cl- ion is favored for reduction, then the water reaction is favored for oxidation producing gaseous oxygen, however experiments shown gaseous chlorine is produced and not oxygen. Although the initial analysis is correct, there is another effect that can happen, known as the overvoltage effect. Additional voltage is sometimes required, beyond the voltage predicted by the Finally, reaction 3 is favorable because it describes the proliferation of OH- ions thus letting a probable reduction of H+ ions less favorable an option. The overall reaction for the process according to the analysis would be the following: As the overall reaction indicates, the concentration of chloride ions is reduced in comparison to OH- ions (whose concentration increases). The reaction also shows the production of gaseous hydrogen, chlorine and aqueous sodium hydroxide. Quantitative electrolysis & Faraday LawsQuantitative aspects of electrolysis were originally developed by Michael Faraday in 1834. Faraday is also credited to have coined the terms electrolyte, electrolysis, among many others while he studied quantitative analysis of electrochemical reactions. Also he was an advocate of the law of conservation of energy. First lawFaraday concluded after several experiments on electrical current in non-spontaneous process, the mass of the products yielded on the electrodes was proportional to the value of current supplied to the cell, the length of time the current existed, and the molar mass of the substance analyzed. In other words, the amount of a substance deposited on each electrode of an electrolytic cell is directly proportional to the quantity of electricity passed through the cell. Below a simplified equation of Faraday's first law: Where,
Second lawFaraday devised the laws of chemical electrodeposition of metals from solutions in 1857. He formulated the second law of electrolysis stating "the amounts of bodies which are equivalent to each other in their ordinary chemical action have equal quantities of electricity naturally associated with them." In other terms, the quantities of different elements deposited by a given amount of electricity are in the ratio of their chemical equivalent weights. An important aspect of the second law of electrolysis is electroplating which together with the first law of electrolysis, has a significant number of applications in the industry, as when used to protect metals to avoid corrosion. ApplicationsThere are various electrochemical processes in both nature and industry, like the fabrication of devices with metals or aleations covered with thin deposits of other metal to prevent these artifacts from having corrosion, the reproduction of objects in copper through electrolysis, and the detection of ingested liquor in drunken drivers through the redox reaction. The nervous impulses in neurons are based on electric charges with sodium ions and the expulsion of potassium ions from the cell; also, the body of an electric eel has cells that, individually, sponsor small electric charges, but together produce a 700-volt potential, enough to kill an human or a horse. See also
References
Categories: Electrochemistry | Physical chemistry |
|||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Electrochemistry". A list of authors is available in Wikipedia. | |||||||||||||





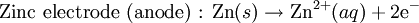
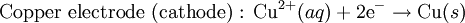
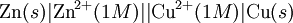
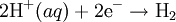
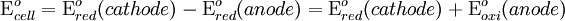
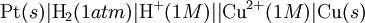
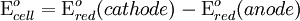
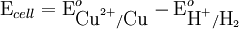
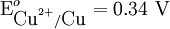
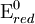 value because the standard electrode potential is an intensive property.
value because the standard electrode potential is an intensive property.
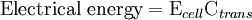
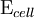 is the cell potential measured in volts (V) and
is the cell potential measured in volts (V) and 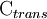 is the cell current integrated over time and measured in coulumbs (C).
is the cell current integrated over time and measured in coulumbs (C). 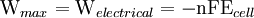
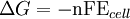
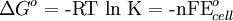
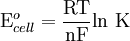
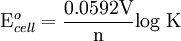
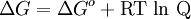 ,
,
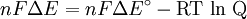
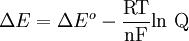
 ) and R =
) and R = 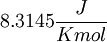 the equation above can be expressed on Base—10 logarithm as shown below:
the equation above can be expressed on Base—10 logarithm as shown below:
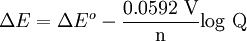
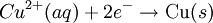
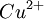 ions increases. Reduction will take place in the cell's compartment where concentration is higher and oxidation will occur on the more dilute side.
ions increases. Reduction will take place in the cell's compartment where concentration is higher and oxidation will occur on the more dilute side.
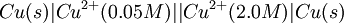
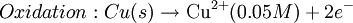
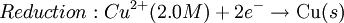
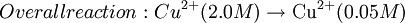
![E = E^{o}- {0.0257 V \over 2} ln {[Cu^{2+}]_{diluted}\over [Cu^{2+}]_{concentrated}}\,](images/math/4/2/a/42a17faf7ff7ef13ea4c4d8884085426.png)
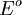 's value of this kind of cell is zero, as electrodes and ions are the same in both half-cells.
After replacing values from the case mentioned, it is possible to calculate cell's potential:
's value of this kind of cell is zero, as electrodes and ions are the same in both half-cells.
After replacing values from the case mentioned, it is possible to calculate cell's potential:
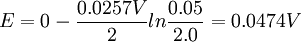
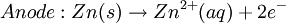
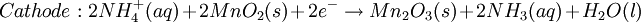
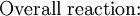
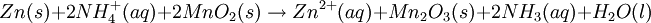
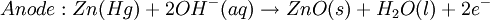
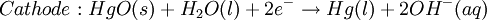
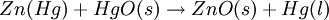
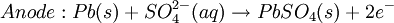
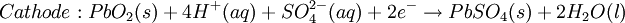
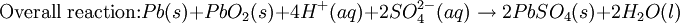
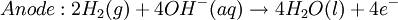
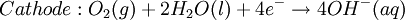
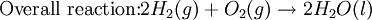
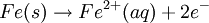
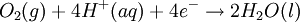
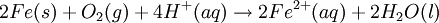
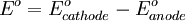
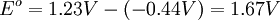
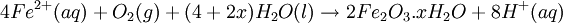
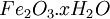 .
The electric circuit works as passage of electrons and ions occurs, thus if an electrolyte is present it will facilitate
.
The electric circuit works as passage of electrons and ions occurs, thus if an electrolyte is present it will facilitate 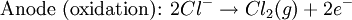
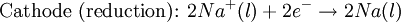
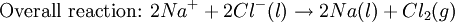
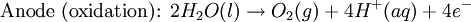
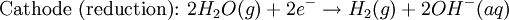
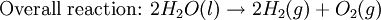
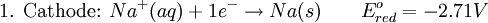
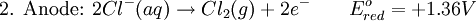
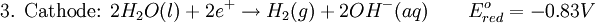
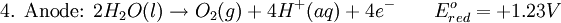
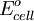 . This may be due to
. This may be due to