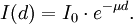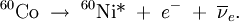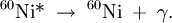To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Gamma ray
Gamma rays (denoted as γ) are a form of electromagnetic radiation or light emission of frequencies produced by sub-atomic particle interactions, such as electron-positron annihilation or radioactive decay. Gamma rays are generally characterized as electromagnetic radiation having the highest frequency and energy, and also the shortest wavelength, within the electromagnetic spectrum, i.e. high energy photons. Due to their high energy content, they can cause serious damage when absorbed by living cells. Product highlight
PropertiesShieldingShielding gamma rays requires large amounts of mass. They are better absorbed by materials with high atomic numbers and high density. The higher the energy of the gamma rays, the thicker the shielding required. Materials for shielding gamma rays are typically measured by the thickness required to reduce the intensity of the gamma rays by one half (the half value layer or HVL). For example, gamma rays that require 1 cm (0.4 inches) of lead to reduce their intensity by 50% will also have their intensity reduced in half by 6 cm (2½ inches) of concrete or 9 cm (3½ inches) of packed dirt.
Matter interaction
When a gamma ray passes through matter, the probability for absorption in a thin layer is proportional to the thickness of that layer. This leads to an exponential decrease of intensity with thickness. The exponential absorption holds only for a narrow beam of gamma rays. If a wide beam of gamma rays passes through a thick slab of concrete, the scattering from the sides reduces the absorption. Here, μ = nσ is the absorption coefficient, measured in cm−1, n the number of atoms per cm3 in the material, σ the absorption cross section in cm2 and d the thickness of material in cm. In passing through matter, gamma radiation ionizes via three main processes: the photoelectric effect, Compton scattering, and pair production.
The secondary electrons (and/or positrons) produced in any of these three processes frequently have enough energy to produce much ionization themselves. Gamma decayGamma rays are often produced alongside other forms of radiation such as alpha or beta. When a nucleus emits an α or β particle, the daughter nucleus is sometimes left in an excited state. It can then jump down to a lower level by emitting a gamma ray in much the same way that an atomic electron can jump to a lower level by emitting visible light or ultraviolet radiation. Gamma rays, x-rays, visible light, and UV rays are all forms of electromagnetic radiation. The only difference is the frequency and hence the energy of the photons. Gamma rays are the most energetic. An example of gamma ray production follows. First 60Co decays to excited 60Ni by beta decay: Then the 60Ni drops down to the ground state (see nuclear shell model) by emitting two gamma rays in succession: Gamma rays of 1.17 MeV and 1.33 MeV are produced. Another example is the alpha decay of 241Am to form 237Np; this alpha decay is accompanied by gamma emission. In some cases, the gamma emission spectrum for a nucleus (daughter nucleus) is quite simple, (eg 60Co/60Ni) while in other cases, such as with (241Am/237Np and 192Ir/192Pt), the gamma emission spectrum is complex, revealing that a series of nuclear energy levels can exist. The fact that an alpha spectrum can have a series of different peaks with different energies reinforces the idea that several nuclear energy levels are possible. Because a beta decay is accompanied by the emission of a neutrino which also carries energy away, the beta spectrum does not have sharp lines, but instead is a broad peak. Hence from beta decay alone it is not possible to probe the different energy levels found in the nucleus. In optical spectroscopy, it is well known that an entity which emits light can also absorb light at the same wavelength (photon energy). For instance, a sodium flame can emit yellow light as well as absorb the yellow light from a sodium vapour lamp. In the case of gamma rays, this can be seen in Mössbauer spectroscopy. Here, a correction for the energy lost by the recoil of the nucleus is made and the exact conditions for gamma ray absorption through resonance can be attained. This is similar to the Franck Condon effects seen in optical spectroscopy. UsesBecause the wavelength of gamma radiation is so short, a single incident photon can impart significant damage to a living cell. This property means that gamma radiation is often used to kill living organisms, in a process called irradiation. Applications of this include sterilising medical equipment (as an alternative to autoclaves or chemical means), removing decay-causing bacteria from many foodstuffs or preventing fruit and vegetables from sprouting to maintain freshness and flavour. Due to their tissue penetrating property, gamma rays/X-rays have a wide variety of medical uses such as in CT Scans and radiation therapy (see X-ray). However, as a form of ionizing radiation they have the ability to effect molecular changes, giving them the potential to cause cancer when DNA is affected. The molecular changes can also be used to alter the properties of semi-precious stones, and is often used to change white topaz into blue topaz. Despite their cancer-causing properties, gamma rays are also used to treat some types of cancer. In the procedure called gamma-knife surgery, multiple concentrated beams of gamma rays are directed on the growth in order to kill the cancerous cells. The beams are aimed from different angles to focus the radiation on the growth while minimizing damage to the surrounding tissues. Gamma rays are also used for diagnostic purposes in nuclear medicine. Several gamma-emitting radioisotopes are used, one of which is technetium-99m. When administered to a patient, a gamma camera can be used to form an image of the radioisotope's distribution by detecting the gamma radiation emitted. Such a technique can be employed to diagnose a wide range of conditions (e.g. spread of cancer to the bones). Gamma ray detectors are also starting to be used in Pakistan as part of the Container Security Initiative (CSI). These US$5 million machines are advertised to scan 30 containers per hour. The objective of this technique is to pre-screen merchant ship containers before they enter U.S. ports. Health effectThe gamma rays are the most dangerous form of radiation emitted by a nuclear explosion because of the difficulty in stopping them. They are also the smallest ray in the electromagnetic spectrum. Gamma-rays are not stopped by the skin. They can induce DNA alteration by interfering with the genetic material of the cell. DNA double-strand breaks are generally accepted to be the most biologically significant lesion by which ionizing radiation causes cancer and hereditary disease.[1]. A study done on Russian nuclear workers exposed to external whole-body gamma radiation at high cumulative doses shows the link between radiation exposure and death from leukemia, lung, liver, skeletal and other solid cancers.[2]. Alongside radiation, gamma-rays also produce thermal burn injuries and induce an immunosuppressive effect.[3][4] Body responseAfter gamma-irradiation, and the breaking of DNA double-strands, a cell can repair the damaged genetic material to the limit of its capability[citation needed]. However, a study of Rothkamm and Lobrich has shown that the repairing process works well after high-dose exposure but is much slower in the case of a low-dose exposure. [5] This could mean that a chronic low-dose exposure cannot be fought by the body[citation needed]. The probability of detecting small alterations or of a detectable defect occurring is most likely small enough that the cell would replicate before initiating a full repair[citation needed]. Some cells can not detect their own genetic defects[citation needed]. Risk assessmentThe natural outdoor exposure in Great Britain is in the range 20-40 nSv/h.[6] Natural exposure to gamma rays is about 1 to 2 mSv a year, and the average total amount of radiation received in one year per inhabitant in the USA is 3.6 mSv.[7] By comparison, the radiation dose from chest radiography is a fraction of the annual naturally occurring background radiation dose,[8] and the dose from fluoroscopy of the stomach is, at most, 0.05 Sv on the skin of the back. For acute full-body equivalent dose, 1 Sv causes slight blood changes, 2-5 Sv causes nausea, hair loss, hemorrhaging and will cause death in many cases. More than 3 Sv will lead to death in less than two months in more than 80 percent of cases, and much over 4 Sv usually causes death (see Sievert).[clarify][citation needed] For low dose exposure, for example among nuclear workers, who receive an average radiation dose of 19mSv, the risk of dying from cancer (excluding leukemia) increases by 2 percent. For a dose of 100mSv, that risk increase is at 10 percent. By comparison, it was 32 percent for the Atom Bomb survivors.[9]. References
See also
Categories: Electromagnetic spectrum | Radioactivity | IARC Group 1 carcinogens |
|||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Gamma_ray". A list of authors is available in Wikipedia. | |||||||||||||||||||||||||||