To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
1s Slater-type function
A normalized 1s Slater-type function is a function which has the form The parameter ζ is called the Slater orbital exponent. Related sets of functions can be used to construct STO-nG basis sets which are used in quantum chemistry. Product highlight
Applications for hydrogen-like atomic systemsA hydrogen-like atom or a hydrogenic atom is an atom with one electron. Except for the hydrogen atom itself (which is neutral) these atoms carry positive charge Exact energy of a hydrogen-like atomThe energy of a hydrogenic system can be exactly calculated analytically as follows :
Integrals needed
The optimum value for Non relativistic energyThe following energy values are thus calculated by using the expressions for energy and for the Slater exponent.
Relativistic energy of Hydrogenic atomic systemsHydrogenic atomic systems are suitable models to demonstrate the relativistic effects in atomic systems in a simple way. The energy expectation value can calculated by using the Slater orbitals with or without considering the relativistic correction for the Slater exponent
|
Notes
- ^ Attila Szabo and Neil S. Ostlund (1996). Modern Quantum Chemistry - Introduction to Advanced Electronic Structure Theory. Dover Publications Inc., 153. ISBN 0486691861.
- ^ In quantum chemistry an orbital is synonymous with "one-electron function", i.e., a function of x, y, and z.


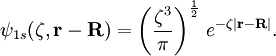



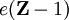 , where
, where 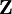 is the
is the 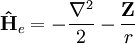 , where
, where 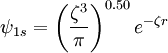 , where
, where  is the Slater exponent.
is the Slater exponent.
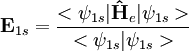 , where
, where 
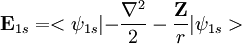
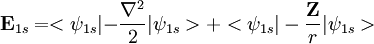
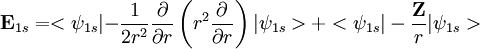 . Using the expression for Slater orbital,
. Using the expression for Slater orbital, ![\mathbf E_{1s} = <\left (\frac{\zeta^3}{\pi} \right ) ^{0.50}e^{-\zeta r}|-\left (\frac{\zeta^3}{\pi} \right )^{0.50}e^{-\zeta r}\left[\frac{-2r\zeta+r^2\zeta^2}{2r^2}\right]>+<\psi_{1s}| - \frac{\mathbf Z}{r}|\psi_{1s}>](images/math/1/1/b/11b9a1c2c366e96d4cfdfddd604e76c2.png)
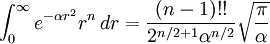 when 'n' is even.
when 'n' is even.
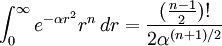 when 'n' is odd.
when 'n' is odd.
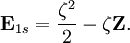
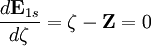 . Thus
. Thus 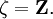
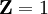 and
and 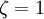
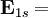 -0.5 hartree
-0.5 hartree
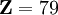 and
and 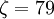
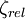 is given as
is given as
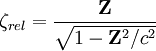 .
.
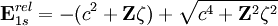 .
.
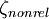
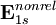
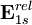 using
using 

