To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Chemical shiftIn nuclear magnetic resonance (NMR), the chemical shift describes the dependence of nuclear magnetic energy levels on the electronic environment in a molecule.[1][2][3] Chemical shifts are relevant in NMR spectroscopy techniques such proton NMR and carbon-13 NMR. An atomic nucleus can have a magnetic moment (nuclear spin), which gives rise to different energy levels and resonance frequencies in a magnetic field. The total magnetic field experienced by a nucleus includes local magnetic fields induced by currents of electrons in the molecular orbitals (note that electrons have a magnetic moment themselves). The electron distribution of the same type of nucleus (e.g. 1H, 13C, 15N) usually varies according to the local geometry (binding partners, bond lengths, angles between bonds, ...), and with it the local magnetic field at each nucleus. This is reflected in the spin energy levels (and resonance frequencies). The variations of nuclear magnetic resonance frequencies of the same kind of nucleus, due to variations in the electron distribution, is called the chemical shift. The size of the chemical shift is given with respect to a reference frequency or reference sample (see also chemical shift referencing), usually a molecule with a barely distorted electron distribution. The chemical shift is of great importance for NMR spectroscopy, a technique to explore molecular properties by looking at nuclear magnetic resonance phenomena. Product highlight
Chemical shift referencingThe chemical shift δ is expressed quantitatively as the difference in NMR frequencies between the site of interest and a reference signal (νX and νref respectively) relative to the operating frequency, ν0 (the NMR frequency of the nuclide at that magnetic field): Since the numerator is usually of the order of hertz, and the denominator of the order of megahertz, δ is generally expressed in parts per millions (ppm). Measuring the NMR frequencies relative to a reference signal means that small drifts in the magnetic field have no effect on the measured values, and expressing the frequency differences in fractional terms means that the δ values are independent of the magnetic field used. In other words, although differences in NMR frequencies (and hence resolution) vary with applied magnetic field, chemical shift values remain constant. The primary reference for 1H, 13C, and 29Si NMR is TMS (tetramethylsilane), which is assigned the chemical shift of zero. Other materials are used for referencing the chemical shift for other nuclides. It is always possible to use a secondary reference, that is, a compound whose non-zero shift has been determined against the primary reference. Note that current spectrometers often dispense with an explicit reference compound and use the 2 NMR signal from the deuterated solvent as a frequency reference. For example, an NMR signal that is 300 Hz higher than TMS at an operating frequency of 300 MHz has a chemical shift of: The operating frequency for a given nucleus can always be calculated from ν0 = (γ / 2π)B, where γ is the gyromagnetic ratio of the nucleus (see Larmor precession, noting that angular frequencies are being used). For instance, γ / 2π = 42.576 MHz T − 1 for 1H and so the proton operating frequency for a 7 tesla magnet is The induced magnetic fieldThe electrons around a nucleus will circulate in a magnetic field and create a secondary induced magnetic field. This field opposes the applied field as stipulated by Lenz's law and the nucleus is therefore said to be shielded. Trends in chemical shift are explained based on the degree of shielding or deshielding. Nuclei are found to resonate in a wide range to the left (or more rare to the right) of the internal standard. When a signal is found with a higher chemical shift:
Conversely a lower chemical shift is called a diamagnetic shift, and is upfield and more shielded. Factors causing chemical shiftsImportant factors influencing chemical shift are electron density, electronegativity of neighboring groups and anisotropic induced magnetic field effects. Electron density shields a nucleus from the external field. For example in proton NMR the electron-poor tropylium ion has its protons downfield at 9.17 ppm, those of the electron-rich cyclooctatetraenyl anion move upfield to 6.75 ppm and its dianion even more upfield to 5.56 ppm. A nucleus in the vicinity of an electronegative atom experiences reduced electron density and the nucleus is therefore deshielded. In proton NMR of methyl halides (CH3X) the chemical shift of the methyl protons increase in the order I > Br > Cl > F from 2.16 ppm to 4.26 ppm reflecting this trend. In carbon NMR the chemical shift of the carbon nuclei increase in the same order from around -10 ppm to 70 ppm. Also when the electronegative atom is removed further away the effect diminishes until it can be observed no longer. Anisotropic induced magnetic field effects are the result of a local induced magnetic field experienced by a nucleus resulting from circulating electrons that can either be paramagnetic when it is parallel to the applied field or diamagnetic when it is opposed to it. It is observed in alkenes where the double bond is oriented perpendicular to the external field with pi electrons likewise circulating at right angles. The induced magnetic field lines are parallel to the external field at the location of the alkene protons which therefore shift downfield to a 4.5 ppm - 7.5 ppm range. The three-dimensional space where a nucleus experiences diamagnetic shift is called the shielding zone with a cone-like shape aligned with the external field. The protons in aromatic compounds are shifted downfield even further with a signal for benzene at 7.73 ppm as a consequence of a diamagnetic ring current. Alkyne protons by contrast resonate at high field in a 2-3 ppm range. For alkynes the most effective orientation in the external field in parallel with electrons circulation around the triple bond. In this way the acetylenic protons are located in the cone-shaped shielding zone hence the upfield shift. Magnetic properties of most common nuclei1H and 13C aren't the only nuclei susceptible to NMR experiments. A number of different nuclei can also be detected, although the use of such techniques is generally rare due to small relative sensitivities in NMR experiments (compared to 1H) of the nuclei in question, the other factor for rare use being their slender representation in nature/organic compounds.
1H, 13C, 15N, 19F and 31P are the five nuclei that have the greatest importance in NMR experiments:
Other chemical shiftsThe related Knight shift (first reported in 1949) is observed with pure metals. The NMR chemical shift in its present day meaning first appeared in journals in 1950. Chemical shifts with a different meaning appear in X-ray photoelectron spectroscopy as the shift in atomic core-level energy due to a specific chemical environment. The term is also used in Mössbauer spectroscopy [6] See also
References
Categories: Nuclear chemistry | Spectroscopy | Nuclear magnetic resonance |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Chemical_shift". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||





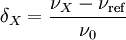
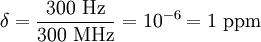
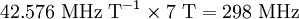 .
.


