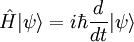To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Davisson–Germer experiment
In physics, the Davisson–Germer experiment provided a critically important confirmation of the de Broglie hypothesis that particles, such as electrons, could behave as waves. More generally, it helped cement the acceptance of quantum mechanics and of Schrödinger's wave equation. Product highlightIn 1927 at Bell Labs, Clinton Davisson and Lester Germer fired slow moving electrons at a crystalline nickel target. The angular dependence of the reflected electron intensity was measured, and was determined to have the same diffraction pattern as those predicted by Bragg for X-rays. This experiment, like Arthur Compton's experiment which gave support to the particle-like nature of light, lent support to de Broglie's hypothesis on the wave-like nature of matter and completed the wave-particle duality hypothesis, which was a fundamental step in quantum theory. |
||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Davisson–Germer_experiment". A list of authors is available in Wikipedia. |