To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Eyring equationThe Eyring equation also known as Eyring-Polanyi equation in chemical kinetics relates the reaction rate to temperature. It was developed almost simultaneously in 1935 by Henry Eyring, M.G. Evans and Michael Polanyi. This equation follows from the transition state theory (aka, activated-complex theory) and contrary to the empirical Arrhenius equation this model is theoretical and based on statistic thermodynamics. Product highlightThe general form of the Eyring-Polanyi equation resemble somewhat to the Arrhenius equation:
It can be rewritten as:
References
Categories: Chemical kinetics | Physical chemistry |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Eyring_equation". A list of authors is available in Wikipedia. |





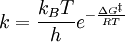
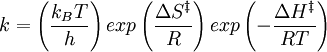
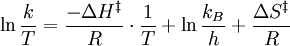
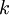 =
= 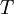 = absolute temperature
= absolute temperature
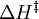 = enthalpy of activation
= enthalpy of activation
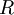 =
= 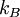 =
= 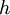 = Planck's constant
= Planck's constant
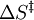 = entropy of activation
= entropy of activation
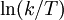 versus
versus 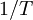 gives a straight line with slope
gives a straight line with slope 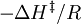 from which the
from which the 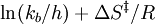 from which the entropy of activation is derived.
from which the entropy of activation is derived.


