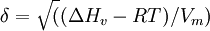To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Hildebrand solubility parameterThe Hildebrand solubility parameter (δ) provides a numerical estimate of the degree of interaction between materials, and can be a good indication of solubility, particularly for non polar materials such as many polymers. Materials with similar values of δ are likely to be miscible. Product highlight
DefinitionThe Hildebrand solubility parameter is the square root of the cohesive energy density:
The cohesive energy density is the amount of energy needed to completely remove unit volume of molecules from their neighbours to infinite separation (an ideal gas), which is equal to the heat of vaporisation divided by molar volume. In order for a material to dissolve, these same interactions need to be overcome as the molecules are separated from each other and surrounded by the solvent. Dr. Joel H. Hildebrand suggested the square root of the cohesive energy density as a numerical value indicating solvency behaviour. This later became known as the “Hildebrand solubility parameter”. Materials with similar solubility parameters will be able to interact with each other, resulting in solvation, miscibility or swelling. Uses and LimitationsIt provides useful predictions for non-polar and slightly polar ( dipole moment <2 debyes) systems without hydrogen bonding. It has found particular use in polymer / solvent interactions, where it provides useful predictions of solubility and swelling of polymers by solvents. More complicated, 3 dimensional solubility parameters, such as Hansen Solubility Parameters can be used for polar molecules.
UnitsConventional units are (calories / cc3)1/2, or cal1/2 cm3/2 SI units are (MPa)1/2 (The unit is one of pressure as one pascal is a joule per cubic meter.) Conversion from the two is by δ (MPa1/2) = 2.0455 δ (cal1/2 cm3/2) Given the non-exact nature of the use of δ, it is often sufficient to say MPa 1/2 is twice (cal1/2 cm3/2). Where the units are not be given, for example in older books, it is usually safe to assume the non SI unit. Examples
From the table, poly(ethylene) has a solubility parameter of 7.9 cal1/2 cm3/2. Good solvents are likely to be diethyl ether and hexane. Poly(styrene) has a solubility parameter of 9.1 cal1/2 cm3/2 , and thus ethyl acetate is likely to be a good solvent. Nylon 6,6 has a solubility parameter of 13.7 cal1/2 cm3/2, and ethanol is likely to be the best solvent of those tabulated. However, the latter is polar, and thus we should be very cautions about using just the Hildebrand solubility parameter to make predictions.
BibliographyBarton, AFM (1991). Handbook of Solubility Parameters and Other Cohesion Parameters, 2nd edition. CRC Press. Barton, AFM (1990). Handbook of Polymer Liquid Interaction Parameters and Other Solubility Parameters. CRC Press. References |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Hildebrand_solubility_parameter". A list of authors is available in Wikipedia. |