To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Inversion temperatureThe inversion temperature in thermodynamics and cryogenics is the critical temperature below which a non-ideal gas (all gases in reality) that is expanded at constant enthalpy will experience a temperature decrease, and above which will experience a temperature increase. This temperature change is known as the Joule-Thomson effect, and is exploited in the liquefaction of gases. Product highlightTheoryThe Joule-Thomson effect cannot be described in the theory of ideal gases, in which interactions between particles are ignored. Instead, one must use a theory that accounts for the Van der Waals force between interacting particles that becomes much stronger as a gas becomes a liquid. For a van der Waals gas we can calculate the enthalpy H using statistical mechanics as
where N is the number of molecules, V is volume, T is temperature (in the Kelvin scale), kB is Boltzmann's constant, and a and b are constants depending on intermolecular forces and molecular volume, respectively. From this equation, we note that if we keep enthalpy constant and increase volume, temperature must change depending on the sign of bkBT − 2a. Therefore, our inversion temperature is given where the sign flips at zero, or
where Tc is the critical temperature of the substance. So for T > Tinv, an expansion at constant enthalpy increases temperature as the work done by the repulsive interactions of the gas is dominant, and so the change in energy is negative. But for T < Tinv, expansion causes temperature to decrease because the work of attractive intermolecular forces dominates, giving a positive change in energy. [1] See also
References
Categories: Thermodynamics | Chemical engineering |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Inversion_temperature". A list of authors is available in Wikipedia. |





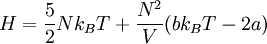 ,
,
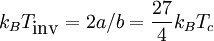 ,
,


