To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Joule-Thomson effect
In physics, the Joule-Thomson effect, or Joule-Kelvin effect, is a process in which the temperature of an ideal gas does not change, but the temperature of a real gas is either decreased or increased by letting the gas expand freely at constant enthalpy (which means that no heat is transferred to or from the gas, and no external work is extracted). It's named for James Prescott Joule and William Thomson, 1st Baron Kelvin who established the effect in 1852 following earlier work by Joule on Joule expansion in which a gas expands at constant internal energy. Product highlight
DescriptionThe relationship between temperature, pressure and volume of a gas is simply described by the various gas laws. When volume is increased in an irreversible process, the gas laws do not uniquely determine what happens to the pressure and temperature of the gas. Isentropic expansion, in which the gas does positive work in the process of expansion, always causes a decrease in temperature. However, when a real gas (as differentiated from an ideal gas) expands freely at constant enthalpy, the temperature may either decrease or increase, depending on the initial temperature and pressure. For any given pressure, a real gas has a Joule-Thomson (Kelvin) inversion temperature, above which expansion at constant enthalpy causes the temperature to rise, and below which expansion at constant enthalpy causes cooling. For most gases at atmospheric pressure, the inversion temperature is fairly high (above room temperature), and so most gases at those temperature and pressure conditions are cooled by isenthalpic expansion. The Joule-Thomson (Kelvin) coefficientThe change of temperature with respect to a change of pressure in a Joule-Thomson process is the Joule-Thomson (Kelvin) coefficient: The value of μJT is typically expressed in °C/bar (SI units: K/Pa) and depends on the specific gas, as well as the temperature and pressure of the gas before expansion. For all real gases, it will equal zero at some point called the inversion point and, as explained above, the Joule-Thomson inversion temperature is the temperature where the coefficient changes sign (i.e., where the coefficient equals zero). The Joule-Thomson inversion temperature depends on the pressure of the gas before expansion. In any gas expansion, the gas pressure decreases and thus the sign of
Helium and hydrogen are two gases whose Joule-Thomson inversion temperatures at one atmosphere are very low (e.g., about −222 °C for helium). Thus, helium and hydrogen will warm when expanded at constant enthalpy at typical room temperatures. On the other hand nitrogen has an inversion temperature of 621 K (348 °C) and oxygen has an inversion temperature of 764 K (491 °C): the two most abundant gases in air can be cooled by the Joule-Thomson effect at typical room temperatures. It should be noted that μJT is always equal to zero for ideal gases (i.e., they will neither heat nor cool upon being expanded at constant enthalpy). Physical mechanismAs a gas expands, the average distance between molecules grows. Because of intermolecular attractive forces, expansion causes an increase in the potential energy of the gas. If no external work is extracted in the process (“free expansion”) and no heat is transferred, the total energy of the gas remains the same because of the conservation of energy. The increase in potential energy thus means a decrease in kinetic energy and therefore in temperature. A second mechanism has the opposite effect. During gas molecule collisions, kinetic energy is temporarily converted into potential energy. As the average intermolecular distance increases, there is a drop in the number of collisions per time unit, which causes a decrease in average potential energy. Again, total energy is conserved, so this leads to an increase in kinetic energy (temperature). Below the Joule-Thompson inversion temperature, the former effect (work done internally against intermolecular attractive forces) dominates, and free expansion causes a decrease in temperature. Above the inversion temperature, the latter effect (reduced collisions causing a decrease in the average potential energy) dominates, and free expansion causes a temperature increase. ApplicationsAs for how the Joule-Thomson effect is achieved in practice:
The effect is applied in the Linde technique as a standard process in the petrochemical industry for example, where the cooling effect is used to liquefy gases, and also in many cryogenic applications (e.g. for the production of liquid oxygen, nitrogen and argon). Only when the Joule-Thomson coefficient for the given gas at the given temperature is greater than zero can the gas be liquefied at that temperature by the Linde cycle. In other words, a gas must be below its inversion temperature to be liquified by the Linde cycle. For this reason, simple Linde cycle liquifiers cannot normally be used to liquify helium, hydrogen and neon. See also
Bibliography
Categories: Thermodynamics | Chemical engineering | Cryogenics | Hydrogen physics |
||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Joule-Thomson_effect". A list of authors is available in Wikipedia. |





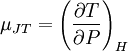
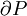 is always negative. With that in mind, the following table explains when the Joule-Thomson effect cools or heats a real gas:
is always negative. With that in mind, the following table explains when the Joule-Thomson effect cools or heats a real gas:
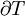 must be
must be

