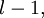To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Magnetic quantum numberIn atomic physics, the magnetic quantum number is the third of a set of quantum numbers (the principal quantum number, the azimuthal quantum number, the magnetic quantum number, and the spin quantum number) which describe the unique quantum state of an electron and is designated by the letter m. The magnetic quantum number denotes the energy levels available within a subshell. Product highlightDerivationThere are a set of quantum numbers associated with the energy states of the atom. The four quantum numbers n, l, m, and s specify the complete and unique quantum state of a single electron in an atom called its wavefunction or orbital. The wavefunction of the Schrödinger wave equation reduces to the three equations that when solved lead to the first three quantum numbers. Therefore, the equations for the first three quantum numbers are all interrelated. The magnetic quantum number arose in the solution of the azimuthal part of the wave equation as shown below. The magnetic quantum number associated with the quantum state is designated as m. The quantum number m refers, loosely, to the direction of the angular momentum vector. The magnetic quantum number m does not affect the electron's energy, but it does affect the probability cloud. Given a particular To describe the magnetic quantum number m you begin with an atomic electron's angular momentum, L, which is related to its quantum number where To show that only certain discrete amounts of angular momentum are allowed, Another way of stating the formula for the magnetic quantum number (ml = Where the quantum number
The magnetic quantum number determines the energy shift of an atomic orbital due to an external magnetic field, hence the name magnetic quantum number (Zeeman effect). However, the actual magnetic dipole moment of an electron in an atomic orbital arrives not only from the electron angular momentum, but also from the electron spin, expressed in the spin quantum number. See also
|
|||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Magnetic_quantum_number". A list of authors is available in Wikipedia. | |||||||||||||||||||||





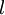 , m is entitled to be any integer from
, m is entitled to be any integer from 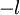 up to
up to 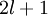 integral magnetic quantum numbers m ranging from
integral magnetic quantum numbers m ranging from 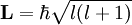
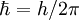 is Planck's reduced constant, also called Dirac's constant. The energy of any wave is the frequency multiplied by Planck's constant. This causes the wave to display particle-like packets of energy called quanta. To show each of the quantum numbers in the quantum state, the formulae for each quantum number include Planck's reduced constant which only allows particular or discrete or quantized energy levels.
is Planck's reduced constant, also called Dirac's constant. The energy of any wave is the frequency multiplied by Planck's constant. This causes the wave to display particle-like packets of energy called quanta. To show each of the quantum numbers in the quantum state, the formulae for each quantum number include Planck's reduced constant which only allows particular or discrete or quantized energy levels.
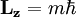
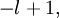 …, 0, …,
…, 0, …,