To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Magnetic susceptibilityIn physics and applied disciplines such as electrical engineering, the magnetic susceptibility is the degree of magnetization of a material in response to an applied magnetic field. Product highlightDefinition of volume susceptibilityThe volume magnetic susceptibility, represented by the symbol where, in SI units,
The magnetic induction B is related to H by the relationship where μ0 is the permeability of free space (see table of physical constants), and
Sometimes [1] an auxiliary quantity, called intensity of magnetization and measured in tesla, is defined as
This allows an alternative description of all magnetization phenomena in terms of the quantities I and B, as opposed to the commonly used M and H. Conversion between SI and cgs unitsNote that these definitions are according to SI conventions. However, many tables of magnetic susceptibility give cgs values that rely on a different definition of the permeability of free space. The cgs value of volume susceptibility is multiplied by 4π to give the SI volume susceptibility value: For example, the cgs volume magnetic susceptibility of water at 20°C is -7.19×10-7 which is -9.04×10-6 using the SI convention. Mass susceptibility and molar susceptibilityThere are two other measures of susceptibility, the mass magnetic susceptibility (χmass or χg, sometimes χm), measured in m3·kg-1 in SI or in cm3·g-1 in cgs and the molar magnetic susceptibility (χmol) measired in m3·mol-1 (SI) or cm3·mol-1 (cgs) that are defined below, where ρ is the density in kg·m-3 (SI) or g·cm-3 (cgs) and M is molar mass in kg·mol-1 (SI) or g·mol-1 (cgs).
Sign of susceptibility: diamagnetics and paramagneticsIf χ is positive, then (1+χv) > 1 (or, in cgs units, (1+4πχv) > 1) and the material is called paramagnetic. In this case, the magnetic field is strengthened by the presence of the material. Alternatively, if χ is negative, then (1+χv) < 1 (or, in cgs units, (1+4πχv) < 1), and the material is diamagnetic. As a result, the magnetic field is weakened in the presence of the material. Experimental methods to determine susceptibilityVolume magnetic susceptibility is measured by the force change felt upon the application of a magnetic field gradient [2]. Early measurements were made using the Gouy balance where a sample is hung between the poles of an electromagnet. The change in weight when the electromagnet is turned on is proportional to the susceptibility. Today, high-end measurement systems use a superconductive magnet. An alternative is to measure the force change on a strong compact magnet upon insertion of the sample. This system, widely used today, is called the Evan's balance. For liquid samples, the susceptibility can be measured from the dependence of the NMR frequency of the sample on its shape or orientation[3][4][5][6][7]. Tensor susceptibilityThe magnetic susceptibility of most crystals is not a scalar. Magnetic response M is dependent upon the orientation of the sample and can occur in directions other than that of the applied field H. In these cases, volume susceptibility is defined as a tensor
where i and j refer to the directions (e.g., x, y and z in Cartesian coordinates) of the applied field and magnetization, respectively. The tensor is thus rank 2, dimension (3,3) describing the component of magnetization in the i-th direction from the external field applied in the j-th direction. Differential susceptibilityIn ferromagnetic crystals, the relationship between M and H is not linear. To accommodate this, a more general definition of differential susceptibility is used where Susceptibility in the frequency domainWhen the magnetic susceptibility is studied as a function of frequency, the permeability is a complex quantity and resonances can be seen. In particular, when an ac-field is applied perpendicular to the detection direction (called the "transverse susceptibility" regardless of the frequency), the effect has a peak at the ferromagnetic resonance frequency of the material with a given static applied field. Currently, this effect is called the microwave permeability or network ferromagnetic resonance in the literature. These results are sensitive to the domain wall configuration of the material and eddy currents. In terms of ferromagnetic resonance, the effect of an ac-field applied along the direction of the magnetization is called parallel pumping. Examples
Sources of confusion in published dataThere are tables of magnetic susceptibility values published on-line that seem to have been uploaded from a substandard source [13], which itself has probably borrowed heavily from the CRC Handbook of Chemistry and Physics. Some of the data (e.g. for Al, Bi, and diamond) are apparently in cgs Molar Susceptibility units, whereas that for water is in Mass Susceptibility units (see discussion above). The susceptibility table in the CRC Handbook is known to suffer from similar errors, and even to contain sign errors. Effort should be made to trace the data in such tables to the original sources, and to double-check the proper usage of units. Use them at your own risk!
See also
Notes
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Magnetic_susceptibility". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||





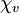 (often simply
(often simply 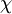 , sometimes
, sometimes 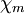 — magnetic, to distinguish from the
— magnetic, to distinguish from the 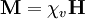
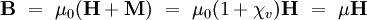
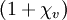 is the relative permeability of the material.
The magnetic susceptibility χv and the magnetic permeability μ are related by the following formula:
is the relative permeability of the material.
The magnetic susceptibility χv and the magnetic permeability μ are related by the following formula:
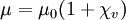 .
.
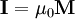 .
.
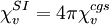
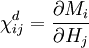
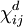 is a tensor derived from partial derivatives of components of M with respect to components of H.
When the
is a tensor derived from partial derivatives of components of M with respect to components of H.
When the 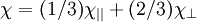 .
. 

