To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
PolarizabilityPolarizability is the relative tendency of a charge distribution, like the electron cloud of an atom or molecule, to be distorted from its normal shape by an external electric field, which may be caused by the presence of a nearby ion or dipole. Product highlightThe electronic polarizability α is defined as the ratio of the induced dipole moment
Polarizability has the SI units of C·m2·V-1 = A2·s4·kg-1 but is more often expressed as polarizabilty volume with units of cm3 or in Å3 = 10-24 cm3.
The polarizability of individual particles is related to the average electric susceptibility of the medium by the Clausius-Mossotti relation. Note that the polarizability α as defined above is a scalar quantity. This implies that the applied electric fields can only produce polarization components parallel to the field. For example, an electric field in the x-direction can only produce an x component in See alsoCategories: Atomic physics | Electric and magnetic fields in matter |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Polarizability". A list of authors is available in Wikipedia. |





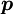 of an atom to the electric field
of an atom to the electric field 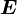 that produces this dipole moment.
that produces this dipole moment.
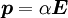
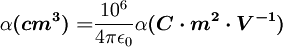 where
where  3 matrix.
3 matrix.


