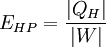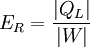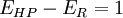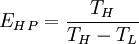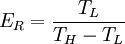To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Thermal efficiencyIn thermodynamics, the thermal efficiency (
From the first and second law of thermodynamics, the output can't exceed what is input, so
When expressed as a percentage, the thermal efficiency must be between 0% and 100%. Due to inefficiencies such as friction, heat loss, and other factors, thermal efficiencies are typically much less than 100%. For example, a typical gasoline automobile engine operates at around 25% thermal efficiency, and a large coal-fueled electrical generating plant peaks at about 36%. The largest diesel engine in the world peaks at 51.7%. In a combined cycle plant thermal efficiencies are approaching 60%. Product highlight
Heat enginesWhen transforming thermal energy into mechanical energy, the thermal efficiency of a heat engine is the percentage of heat energy that is transformed into work. Thermal efficiency is defined as
Carnot efficiencyThe second law of thermodynamics puts a fundamental limit on the thermal efficiency of heat engines. Surprisingly, even an ideal, frictionless engine can't convert anywhere near 100% of its input heat into work. The limiting factors are the temperature at which the heat enters the engine,
Energy conversionFor an energy conversion device like a boiler or furnace, the thermal efficiency is
So, for a boiler that produces 210 kW (or 700,000 BTU/h) output for each 300 kW (or 1,000,000 BTU/h) heat-equivalent input, its thermal efficiency is 210/300 = 0.70, or 70%. This means that the 30% of the energy is lost to the environment. An electric resistance heater has a thermal efficiency of at or very near 100%, so, for example, 1500W of heat are produced for 1500W of electrical input. When comparing heating units, such as a 100% efficient electric resistance heater to an 80% efficient natural gas-fueled furnace, an economic analysis is needed to determine the most cost-effective choice. Heat pumps and RefrigeratorsHeat pumps, refrigerators, and air conditioners, for example, move heat, rather than convert it, so other measures are needed to describe their thermal performance. The common measures are the coefficient of performance (COP), energy efficiency ratio (EER), and seasonal energy efficiency ratio (SEER).
*H=high (temperature/heat source), L=low (temperature/heat source) Energy efficiencyThe 'thermal efficiency' is sometimes called the energy efficiency. In the United States, in everyday usage the SEER is the more common measure of energy efficiency for cooling devices, as well as for heat pumps when in their heating mode. For energy-conversion heating devices their peak steady-state thermal efficiency is often stated, e.g., 'this furnace is 90% efficient', but a more detailed measure of seasonal energy effectiveness is the Annual Fuel Utilization Efficiency (AFUE).[2] See also
References |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Thermal_efficiency". A list of authors is available in Wikipedia. |


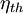 ) is a dimensionless performance measure of a thermal device such as an internal combustion engine, a
) is a dimensionless performance measure of a thermal device such as an internal combustion engine, a 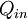 , to the device is
, to the device is 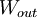 , or heat,
, or heat, 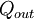 , or possibly both. Because the input heat normally has a real financial cost, a memorable, generic definition of thermal efficiency is
, or possibly both. Because the input heat normally has a real financial cost, a memorable, generic definition of thermal efficiency is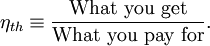
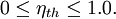



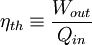
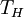 , and the temperature of the environment into which the engine exhausts its waste heat,
, and the temperature of the environment into which the engine exhausts its waste heat,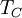 , measured in the absolute
, measured in the absolute 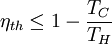
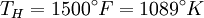 and the ambient temperature is
and the ambient temperature is 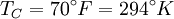 , then its maximum possible efficiency is given by:
, then its maximum possible efficiency is given by:
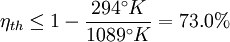
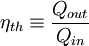 .
.