To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Bragg diffractionThe Bragg formulation of X-ray diffraction (also referred to as Bragg diffraction) was first proposed by William Lawrence Bragg and William Henry Bragg in 1913 in response to their discovery that crystalline solids produced surprising patterns of reflected X-rays (in contrast to that of, say, a liquid). They found that in these crystals, for certain specific wavelengths and incident angles, intense peaks of reflected radiation (known as Bragg peaks) were produced. The concept of Bragg diffraction applies equally to neutron diffraction and electron diffraction processes. W. L. Bragg explained this result by modeling the crystal as a set of discrete parallel planes separated by a constant parameter d. It was proposed that the incident X-ray radiation would produce a Bragg peak if their reflections off the various planes interfered constructively.
Product highlight
MechanicsAs the wave enters the crystal, some portion of it will be reflected by the first layer, while the rest will continue through to the second layer, where the process continues. By the definition of constructive interference, the separately reflected waves will remain in phase if the difference in the path length of each wave is equal to an integer multiple of the wavelength. In the figure 2 on the right, the path difference is given by Waves that satisfy this condition interfere constructively and result in a reflected wave of significant intensity. Reciprocal spaceMore elegant is the description in reciprocal space. Reciprocal lattice vectors describe the set of lattice planes as a normal vector to this plane with length G = 2π / d. Then Bragg's law is simply expressed by the conservation of momentum transfer G = kf − ki with incident and final wave vectors ki and kf of identical length. The precedent relation is also called Laue diffraction and not only gives the absolute value, but a full vectorial description of the phenomenon. The scanning variable can the be the length or the direction of the incident or exit wave vectors relating to energy- and angle-dispersive setups. The simple relationship between diffraction angle and Q-space is then and the unexperienced applicant should just remember larger 2θ equals larger Q.
Selection rules and practical x-ray crystallographyBragg's law, as stated above, can be used to obtain the lattice spacing of a particular cubic system through the following relation: Where a is the lattice spacing of the cubic crystal, and h, k, and l are the Miller indices of the Bragg plane. Combining this relation with Bragg's law: One can derive selection rules for the Miller indices for different cubic Bravais lattices; here, selection rules for several will be given as is.
These selection rules can be used for any crystal with the given crystal structure. Selection rules for other structures can be referenced elsewhere, or derived. Nobel Prize for Bragg diffractionIn 1915, William Henry Bragg and William Lawrence Bragg were awarded the Nobel Prize for their contributions to crystal structure analysis. They were the first and (so far) the only father-son team to have jointly won the prize. Other father/son laureates include Niels and Aage Bohr, Manne and Kai Siegbahn, J.J. and George Thomson, Hans von Euler-Chelpin and Ulf von Euler, and Arthur and Roger Kornberg all having been awarded the prize for separate contributions. W.L. Bragg was 25 years old at the time, making him the youngest Nobel laureate to date. See also
References
Categories: Condensed matter physics | Diffraction |
|||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Bragg_diffraction". A list of authors is available in Wikipedia. |





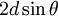 , where d denotes the interplanar distance. This gives the formula for what is known as the Bragg condition or
, where d denotes the interplanar distance. This gives the formula for what is known as the Bragg condition or 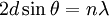
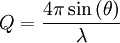
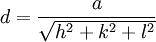
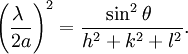
 3n
3n


