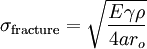To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Fracture
A fracture is the (local) separation of a body into two, or more, pieces under the action of stress. The word fracture is often applied to bones of living creatures, or to crystals or crystalline materials, such as gemstones or metal. Sometimes, in crystalline materials, individual crystals fracture without the body actually separating into two or more pieces. Depending on the substance which is fractured, a fracture reduces strength (most substances) or inhibits transmission of light (optical crystals). A detailed understanding of how fracture occurs in materials may be assisted by the study of fracture mechanics. Product highlight
Types of fractureBrittle fracture
In brittle fracture, no apparent plastic deformation takes place before fracture. In brittle crystalline materials, fracture can occur by cleavage as the result of tensile stress acting normal to crystallographic planes with low bonding (cleavage planes). In amorphous solids, by contrast, the lack of a crystalline structure results in a conchoidal fracture, with cracks proceeding normal to the applied tension. The theoretical strength of a crystalline material is (roughly) where: -
On the other hand, a crack introduces a stress concentration modeled by
where: -
Putting these two equations together, we get Looking closely, we can see that sharp cracks (small ρ) and large defects (large a) both lower the fracture strength of the material. Recently, scientists have discovered supersonic fracture , the phenomenon of crack motion faster than the speed of sound in a material.[citation needed] This phenomenon was recently also verified by experiment of fracture in rubber-like materials. Ductile fractureIn ductile fracture, extensive plastic deformation takes place before fracture. Many ductile metals, especially materials with high purity, can sustain very large deformation of 50–100% or more strain before fracture under favorable loading condition and environmental condition. The strain at which the fracture happens is controlled by the purity of the materials. At room temperature, pure iron can undergo deformation up to 100% strain before breaking, while cast iron or high-carbon steels can barely sustain 3% of strain.[citation needed].
Because ductile rupture involves a high degree of plastic deformation, the fracture behavior of a propagating crack as modeled above changes fundamentally. Some of the energy from stress concentrations at the crack tips is dissipated by plastic deformation before the crack actually propagates. The basic steps of ductile fracture are necking (which results in stress localization at the point on the sample of smallest cross-sectional area), void formation, void coalescence (also known as crack formation), crack propagation, and failure, often resulting in a cup-and-cone shaped failure surface.
Crack Separation ModesThere are three modes of fracture. Mode I, or the opening mode, is characterized by a stress normal to the crack faces. Mode II, the sliding mode or forward shear mode, has a shear stress normal to the crack front. Finally Mode III is the tearing mode, with a shear stress parallel to the crack front. For more information, see fracture mechanics.
See also
Bibliography
|
||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Fracture". A list of authors is available in Wikipedia. | ||||||||||||||





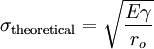
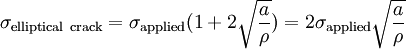 (For sharp cracks)
(For sharp cracks)