To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Indium(III) phosphide
Indium phosphide (InP) is a binary semiconductor composed of indium and phosphorus. It is used in high-power and high-frequency electronics because of its superior electron velocity with respect to the more common semiconductors silicon and gallium arsenide. It also has a direct bandgap, making it useful for optoelectronics devices like laser diodes. Product highlightInP is also used as a substrate for epitaxial indium gallium arsenide based opto-electronic devices. Indium phosphide also has one of the longest-lived optical phonons of any compound with the zincblende crystal structure. Optical propertiesThe Sellmeier equation that describes how the optical refractive index for indium phosphide depends on wavelength is given by
This gives refractive index values rising from around 3.21 at 10 µm and 3.32 at 1.5 µm to 3.47 at 1.0 µm.
ReferencesSuppliers
Categories: Phosphides | Indium compounds | Inorganic compounds | Optoelectronics | Semiconductor materials | III-V compounds |
||||||||||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Indium(III)_phosphide". A list of authors is available in Wikipedia. | ||||||||||||||||||||||||||||||||||||||||||||||





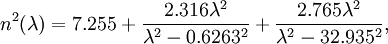 where λ is the wavelength in micrometres.
where λ is the wavelength in micrometres.


