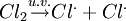To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Radical (chemistry)
In chemistry, radicals (often referred to as free radicals) are atomic or molecular species with unpaired electrons on an otherwise open shell configuration. These unpaired electrons are usually highly reactive, so radicals are likely to take part in chemical reactions. Radicals play an important role in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes, including human physiology. For example, superoxide and nitric oxide regulate many biological processes, such as controlling vascular tone. "Radical" and "free radical" are frequently used interchangeably, however a radical may be trapped within a solvent cage or be otherwise bound. The first organic free radical identified was triphenylmethyl radical, by Moses Gomberg in 1900 at the University of Michigan. Historically, the term radical has also been used for bound parts of the molecule, especially when they remain unchanged in reactions. For example, methyl alcohol was described as consisting of a methyl 'radical' and a hydroxyl 'radical'. Neither were radicals in the usual chemical sense, as they were permanently bound to each other, and had no unpaired, reactive electrons. In mass spectrometry, such radicals are observed after breaking down the substance with a hail of energetic electrons.
Product highlight
Depicting radicals in chemical reactionsIn written chemical equations, free radicals are frequently denoted by a dot placed immediately to the right of the atomic symbol or molecular formula as follows:
Radical reaction mechanisms use single-headed arrows to depict the movement of single electrons: The homolytic cleavage of the breaking bond is drawn with a 'fish-hook' arrow to distinguish from the usual movement of two electrons depicted by a standard curly arrow. It should be noted that the second electron of the breaking bond also moves to pair up with the attacking radical electron; this is not explicitly indicated in this case. In chemistry, free radicals take part in radical addition and radical substitution as reactive intermediates. Chain reactions involving free radicals can usually be divided into three distinct processes: initiation, propagation, and termination.
FormationRadicals may occur naturally as relatively stable chemical compounds. The most common example is molecular dioxygen O2, which in its stable ground state has two unpaired electrons. The formation of radicals may involve breaking of covalent bonds homolytically, a process that requires significant amounts of energy. For example, splitting H2 into 2H· has a ΔH° of +435 kJ/mol, and Cl2 into 2Cl· has a ΔH° of +243 kJ/mol. This is known as the homolytic bond dissociation energy, and is usually abbreviated as the symbol DH°. The bond energy between two covalently bonded atoms is affected by the structure of the molecule as a whole, not just the identity of the two atoms, and radicals requiring more energy to form are less stable than those requiring less energy. Homolytic bond cleavage most often happens between two atoms of similar electronegativity. In organic chemistry this is often the O-O bond in peroxide species or O-N bonds. However, propagation is a very exothermic reaction. Note that most species are electrically neutral although radical ions do exist. Radicals may also be formed by single electron oxidation or reduction of an atom or molecule. An example is the production of superoxide by the electron transport chain. Persistence and stabilityLong lived radicals can be placed into two categories
ReactivityRadical alkyl intermediates are stabilized by similar criteria as carbocations: the more substituted the radical center is, the more stable it is. This will direct their reactions: formation of a tertiary radical (R3C·) is favored over secondary (R2HC·) or primary (RH2C·). However, radicals next to functional groups, such as carbonyl, nitrile, and ether are even more stable than tertiary alkyl radicals. Radicals attack double bonds, but unlike similar ions, they are slightly less directed by electrostatic interactions. For example, the reactivity of nucleophilic ions with α,β-unsaturated compounds (C=C-C=O) is directed by the electron-withdrawing effect of the oxygen, resulting in a partial positive charge on the carbonyl carbon. There are two reactions that are observed in the ionic case: the carbonyl is attacked in a direct addition to carbonyl, or the vinyl is attacked in conjugate addition, and in either case, the charge on the nucleophile is taken by the oxygen. Radicals add rapidly to the double bond, and the resulting α-radical carbonyl is relatively stable. Nonetheless, the electrophilic/neutrophilic character of radicals has been shown in a variety of instances (e.g., in the alternating tendency of the copolymerization of malieic anhydride (electrophilic) and styrene (slightly nuecleophilic). In intramolecular reactions, precise control can be achieved despite the extreme reactivity of radicals. Radicals will attack the closest reactive site the most readily. Therefore, when there is a choice, a preference for five-membered rings is observed: four-membered rings are too strained, and collisions with carbons five or more atoms away in the chain are infrequent. Combustion
Probably the most familiar free-radical reaction for most people is combustion. The oxygen molecule is a stable diradical, best represented by ·O-O·, which is stable because the spins of the electrons are parallel. The ground state of oxygen is an unreactive spin-paired (triplet) radical, but an extremely reactive spin-unpaired (singlet) radical is available. In order for combustion to occur, the energy barrier between these must be overcome. This barrier can be overcome by heat, requiring high temperatures, or can be lowered by enzymes to initiate reactions at the temperatures inside living things. Combustion is comprised of various radical chain reactions that the singlet radical can initiate. The flammability of a given material is strongly dependent on the concentration of free radicals that must be obtained before initiation and propagation reactions dominate leading to combustion of the material. Once the combustible material has been consumed, termination reactions again dominate and the flame dies out. Propagation or termination reactions can be promoted to alter flammability. Tetraethyl lead was once commonly added to gasoline, because it very easily breaks up into radicals, which consume other free radicals in the gasoline-air mixture. This prevents the combustion from initiating prematurely (see engine knock). PolymerizationIn addition to combustion, many polymerization reactions involve free radicals. As a result many plastics, enamels, and other polymers are formed through radical polymerization. Recent advances in radical polymerization methods, known as Living Radical Polymerization, include:
These methods produce polymers with a much narrower distribution of molecular weights. Atmospheric radicalsThe most common radical in the lower atmosphere is molecular dioxygen. In the upper atmosphere free radicals are produced through photodissociation of the source molecules, particularly the normally unreactive chlorofluorocarbons by solar ultraviolet radiation or by reactions with other stratospheric constituents. These free radicals then react with ozone in a catalytic chain reaction which destroys the ozone, but regenerates the free radical, allowing it to participate in additional reactions. Such reactions are believed to be the primary cause of depletion of the ozone layer and this is why the use of chlorofluorocarbons as refrigerants has been restricted. Free radicals in biologyFree radicals play an important role in a number of biological processes, some of which are necessary for life, such as the intracellular killing of bacteria by neutrophil granulocytes. Free radicals have also been implicated in certain cell signalling processes [3]. The two most important oxygen-centered free radicals are superoxide and hydroxyl radical. They are derived from molecular oxygen under reducing conditions. However, because of their reactivity, these same free radicals can participate in unwanted side reactions resulting in cell damage. Many forms of cancer are thought to be the result of reactions between free radicals and DNA, resulting in mutations that can adversely affect the cell cycle and potentially lead to malignancy. Some of the symptoms of aging such as atherosclerosis are also attributed to free-radical induced oxidation of many of the chemicals making up the body. In addition free radicals contribute to alcohol-induced liver damage, perhaps more than alcohol itself. Radicals in cigarette smoke have been implicated in inactivation of alpha 1-antitrypsin in the lung. This process promotes the development of emphysema. Free radicals may also be involved in Parkinson's disease, senile and drug-induced deafness, schizophrenia, and Alzheimer's. The classic free-radical syndrome, the iron-storage disease hemochromatosis, is typically-associated with a constellation of free-radical-related symptoms including movement disorder, psychosis, skin pigmentary melanin abnormalities, deafness, arthritis, and diabetes mellitus. The free radical theory of aging proposes that free radicals underlie the aging process itself, whereas the process of mitohomesis suggests that repeated exposure to free radicals may extend life span. Because free radicals are necessary for life, the body has a number of mechanisms to minimize free radical induced damage and to repair damage which does occur, such as the enzymes superoxide dismutase, catalase, glutathione peroxidase and glutathione reductase. In addition, antioxidants play a key role in these defense mechanisms. These are often the three vitamins, vitamin A, vitamin C and vitamin E and polyphenol antioxidants. Further, there is good evidence bilirubin and uric acid can act as antioxidants to help neutralize certain free radicals. Bilirubin comes from the breakdown of red blood cells' contents, while uric acid is a breakdown product of purines. Too much bilirubin, though, can lead to jaundice, which could eventually damage the central nervous system, while too much uric acid causes gout [4]. Reactive oxygen speciesReactive oxygen species or ROS are species such as superoxide, hydrogen peroxide, and hydroxyl radical and are associated with cell damage. ROSs form as a natural byproduct of the normal metabolism of oxygen and have important roles in cell signaling. Loose definition of radicalsIn most fields of chemistry, the historical definition of radicals contends that the molecules have nonzero spin. However in fields including spectroscopy, chemical reaction, and astrochemistry, the definition is slightly different. Gerhard Herzberg, who won the Nobel prize for his research of electronic structure and geometry of radicals, suggested a looser definition of free radicals: "any transient (chemically unstable) species (atom, molecule, or ion)"[5]. The main point of his suggestion is that there are many chemically unstable molecules which have zero spin, such as C2, C3, CH2 and so on. This definition is more convenient for discussions of transient chemical processes and astrochemistry; therefore researchers in these fields prefer to use this loose definition.[6] DiagnosticsFree Radical diagnostic techniques include:
See alsoReferences
Categories: Chemical bonding | Environmental chemistry | Biomolecules | Free radicals |
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Radical_(chemistry)". A list of authors is available in Wikipedia. |