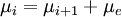To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Saha ionization equationThe Saha ionization equation, also known as the Saha-Langmuir equation, was developed by the Indian astrophysicist Megh Nad Saha in 1920, and later (1923) by Irving Langmuir. One of the important applications of the equation was in explaining the spectral classification of stars. The equation is a result of combining ideas of quantum mechanics and statistical mechanics. Product highlightFor a gas at a high enough temperature, the thermal collisions of the atoms will ionize some of the atoms. One or more of the electrons that are normally bound to the atom in orbits around the atomic nucleus will be ejected from the atom and will form an electron gas that co-exists with the gas of atomic ions and neutral atoms. This state of matter is called a plasma. The Saha equation describes the degree of ionization of this plasma as a function of the temperature, density, and ionization energies of the atoms. The Saha equation only holds for weakly ionized plasmas for which the Debye length is large. This means that the "screening" of the coulomb charge of ions and electrons by other ions and electrons is negligible. The subsequent lowering of the ionization potentials and the "cutoff" of the partition function is therefore also negligible. For a gas composed of a single atomic species, the Saha equation is written: where:
In the case where only one level of ionization is important, we have n1 = ne and defining the total density n as n = n0 + n1, the Saha equation simplifies to: where ε is the energy of ionization. The Saha equation is useful for determining the ratio of particle densities for two different ionization levels. The most useful form of the Saha equation for this purpose is
where Z denotes the partition function. The Saha equation can be seen as a restatement of the equilibrium condition for the chemical potentials: This equation simply states that the potential for an atom of ionization state i to ionize is the same as the potential for an electron and an atom of ionization state i+1; the potentials are equal, therefore the system is in equilibrium and no net change of ionization will occur. |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Saha_ionization_equation". A list of authors is available in Wikipedia. |





![\frac{n_{i+1}n_e}{n_i} = \frac{2}{\Lambda^3}\frac{g_{i+1}}{g_i}\exp\left[-\frac{(\epsilon_{i+1}-\epsilon_i)}{k_BT}\right]](images/math/2/4/8/2484702b34c60d1531ca30548fc34e19.png)
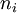 is the density of atoms in the i-th state of ionization, that is with i electrons removed.
is the density of atoms in the i-th state of ionization, that is with i electrons removed.
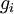 is the degeneracy of states for the i-ions
is the degeneracy of states for the i-ions
 is the energy required to remove i electrons from a neutral atom, creating an i-level ion.
is the energy required to remove i electrons from a neutral atom, creating an i-level ion.
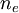 is the electron density
is the electron density
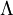 is the
is the 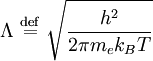
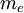 is the mass of an electron
is the mass of an electron
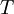 is the
is the 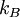 is the
is the 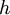 is Planck's constant
is Planck's constant
![\frac{n_e^2}{n-n_e} = \frac{2}{\Lambda^3}\frac{g_1}{g_0}\exp\left[\frac{-\epsilon}{k_BT}\right]](images/math/4/2/7/427932d7b2eda4f33f7bb24517ff820c.png)
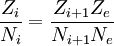 ,
,