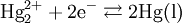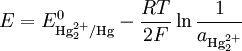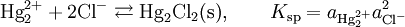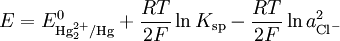To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Saturated calomel electrodeThe Saturated calomel electrode (SCE) is a reference electrode based on the reaction between elemental mercury and mercury(I) chloride. The aqueous phase in contact with the mercury and the mercury(I) chloride (Hg2Cl2, "calomel") is a saturated solution of potassium chloride in water. The electrode is normally linked via a porous frit to the solution in which the other electrode is immersed. This porous frit is a salt bridge. Product highlightIn cell notation the electrode is written as: Cl- (4M)|Hg2Cl2(s)|Hg(l)|Pt
Theory of operationThe electrode is based on the redox reaction The Nernst equation for this reaction is where E0 is the standard electrode potential for the reaction and aHg is the activity for the mercury cation (the activity for a liquid is 1). This activity can be found from the solubility product of the reaction By replacing the activity in the Nernst equation with the value in the solubility equation, we get The only variable in this equation is the activity (or concentration) of the chloride anion. But since the inner solution is satured with potassium chloride, this activity is fixed by the solubility of potassium chloride. At standard conditions, the potential of the saturated calomel electrode should be +0.241 V versus the SHE. ApplicationThe SCE is used in pH measurement, cyclic voltammetry and general aqueous electrochemistry. This electrode and the silver/silver chloride reference electrode work in the same way. In both electrodes, the activity of the metal ion is fixed by the solubility of the metal salt. The calomel electrode contains mercury, which poses much greater health hazards than the silver metal used in the Ag/AgCl electrode. Categories: Electrodes | Electrochemistry |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Saturated_calomel_electrode". A list of authors is available in Wikipedia. |