To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Sedimentation
Sedimentation describes the motion of molecules in solutions or particles in suspensions in response to an external force such as gravity, centrifugal force or electric force. Sedimentation may pertain to objects of various sizes, ranging from suspensions of dust and pollen particles to cellular suspensions to solutions of single molecules such as proteins and peptides. Even small molecules such as aspirin can be sedimented, although it can be difficult to apply a sufficiently strong force to produce significant sedimentation. Product highlightIn a sedimentation experiment, the applied force accelerates the particles to a terminal velocity vterm at which the applied force is exactly canceled by an opposing drag force. In general, the drag force varies linearly with the terminal velocity, i.e., Fdrag = fvterm where f depends only on the properties of the particle and the surrounding fluid. Similarly, the applied force generally varies linearly with some coupling constant (denoted here as q) that depends only on the properties of the particle, Fapp = qEapp. Hence, it is generally possible to define a sedimentation coefficient In many cases, the motion of the particles is blocked by a hard boundary; the resulting accumulation of particles at the boundary is called a sediment. The concentration of particles at the boundary is opposed by the diffusion of the particles. The sedimentation of particles under gravity is described by the Mason-Weaver equation, which has a simple exact solution. The sedimentation coefficient s in this case equals mb / f, where mb is the buoyant mass. The sedimentation of particles under the centrifugal force is described by the Lamm equation, which likewise has an exact solution. The sedimentation coefficient s also equals mb / f, where mb is the buoyant mass. However, the Lamm equation differs from the Mason-Weaver equation because the centrifugal force depends on radius from the origin of rotation, whereas gravity is presumed constant. The Lamm equation also has extra terms, since it pertains to sector-shaped cells, whereas the Mason-Weaver equation pertains to box-shaped cells (i.e., cells whose walls are aligned with the three Cartesian axes). Particles with a charge or dipole moment can be sedimented by an electric field or electric field gradient, respectively. These processes are called electrophoresis and dielectrophoresis, respectively. For electrophoresis, the sedimentation coefficient corresponds to the particle charge divided by its drag (the electrophoretic mobility). Similarly, for dielectrophoresis, the sedimentation coefficient equals the particle's electric dipole moment divided by its drag. See also
Categories: Laboratory techniques | Separation processes |
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Sedimentation". A list of authors is available in Wikipedia. |





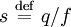 that depends only on the properties of the particle and the surrounding fluid. Thus, measuring s can reveal underlying properties of the particle.
that depends only on the properties of the particle and the surrounding fluid. Thus, measuring s can reveal underlying properties of the particle.


