To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Time-of-flight mass spectrometryTime-of-flight mass spectrometry (TOFMS) is method of mass spectrometry in which ions are accelerated by an electric field of known strength.[1] This acceleration results in an ion having the same kinetic energy as any other ion that has the same charge. The velocity of the ion depends on the mass-to-charge ratio. The time that it subsequently takes for the particle to reach a detector at a known distance is measured. This time will depend on the mass-to-charge ratio of the particle (heavier particles reach lower speeds). From this time and the known experimental parameters one can find the mass-to-charge ratio of the ion. Product highlight
TheoryIt is well understood in physics that the potential energy of a charged particle in an electric field is related to its charge and to the strength of the electric field:
where Ep is potential energy, q is the charge of the particle, and U is the electric potential difference (also known as voltage). When the charged particle is accelerated into time-of-flight tube by the voltage U, its potential energy is converted to kinetic energy. The kinetic energy of any mass is:
In effect, the potential energy is converted to kinetic energy, meaning that equations [1] and [2] are equal
The velocity of the charged particle after acceleration will not change since it moves in a field-free time-of-flight tube. The velocity of the particle can be determined in a time-of-flight tube since the length of the path (d) of the flight of the ion is known and the time of the flight of the ion (t) can be measured using a transient digitizer or time to digital converter. Thus,
and we substitute the value of v in Eqn [5] into Eqn [4].
Rearranging Eqn [6] so that the flight time is expressed by everything else:
Taking the square root of the time
These factors for the time of flight have been grouped purposely.
where k is a proportionality constant representing factors related to the instrument settings and characteristics. Eqn [9] reveals more clearly that the time of flight of the ion varies with the square root of its mass-to-charge ratio (m/q). Consider a real world example of a MALDI time-of-flight mass spectrometer instrument which is used to produce a mass spectrum of the tryptic peptides of a protein. Suppose the mass of one tryptic peptide is 1000 daltons (Da). The kind of ionization of peptides produced by MALDI is typically +1 ions, so q = e in both cases. Suppose the instrument is set to accelerate the ions in a U = 15,000 volts (15 kilovolt, or 15 kV) potential. And suppose the length of the flight tube is 1.5 meters (typical). All the factors necessary to calculate the time of flight of the ions are now known for Eqn [8], which is evaluated first of the ion of mass 1000 Da:
Note that the mass had to be converted from daltons (Da) to kilograms (kg) to make it possible to evaluate the equation in the proper units. The final value should be in seconds: which is about 28 microseconds. If there were a singly charged tryptic peptide ion with 4000 Da mass, and it is four times larger than the 1000 Da mass, it would take twice the time, or about 56 microseconds to traverse the flight tube, since time is proportional to the square root of the mass-to-charge ratio. Delayed extractionResolution can be improved in MALDI-TOF by allowing the initial burst of ions and neutrals produced by the laser pulse to equilibrate and dissipate before the ions are accelerated into the flight tube. This is referred to as "time-lag focusing" [2] for ionization of a gas and "delayed extraction" [3] for desorption ionization. Delayed extraction refers to delaying the onset of the extraction potential by some short time after the ionization event. This differs from leaving the extraction potential, which is responsible for accelerating the ions into the flight tube, continuously on such that the ions are accelerated instantaneously upon being formed. Delayed extraction is often used with MALDI ion sources or other situations where the ions to be analyzed are produced in an expanding plume or sphere. Since spatial homogeneity is important to mass resolution, on first inspection it can appear counter-intuitive to allow the ion plume or sphere to further expand before extraction. Delayed extraction is more of a correction to initial momentum and spatial homogeneity is only critical at the detector. The ions that have developed the greatest momentum away from the direction of extraction are exposed to the accelerating extraction potential for longer due to being further from the extraction plate when the extraction potential is turned on. Conversely those ions that have forward momentum are accelerated less since they start being accelerated at lower potential closer to the extraction plate. In this way the delay time acts as a one-dimensional focusing element. The ions at the back of the distribution will be accelerated more and the ions at the front of the distribution will be accelerated less and when properly adjusted they will be spatially focused at the detector plate. Reflectron TOFThe kinetic energy distribution in the direction of ion flight can be corrected by using a reflectron.[4] The reflectron uses an electrostatic field to reflect the ion beam toward the detector. The more energetic ions penetrate deeper into the reflectron, and take a slightly longer path to the detector. Less energetic ions of the same charge and mass only will penetrate a short distance into the reflectron and take a shorter path the detector. The detector is placed at the focal point where ions of different energies focused by the reflectron strike the detector at the same time. A reflectron TOF uses a similar concept as the delayed extraction technique but at a point half-way through the flight path (where the detector might go in a linear TOF). This is achieved by turning the ions around ~180° by a fixed potential gradient. The ions with more kinetic energy will penetrate deeper into the potential gradient and take longer to turn around than ions with less kinetic energy. The distribution of ions will be re-focused at some further distance. An additional advantage to the reflectron arrangement is that twice the flight path is achieved in a given length of instrument. Ion gatingHigh resolution time-of-flight mass spectrometry requires tight focusing of the ion beam in space, time, and energy. Even an efficient ion gate does not completely correct for velocity distributions originating in the ion source. Orthogonal acceleration time-of-flightContinuous ion sources (most commonly electrospray ionization) are generally interfaced to the TOF mass analyzer by "orthogonal extraction" in which the ions are introduced into the mass analyzer in a direction perpendicular to the direction of flight.[5] This improves duty cycle and thereby sensitivity by analyzing a "slice" of ions instead of a "packet" and removes problems arising from the initial kinetic energy distribution in the axis perpendicular to the direction of ion flight. The ion beam entering the orthogonal extraction region is focused to a small diameter by RF ion guides and electrostatic lenses to minimize the spatial distribution of the beam in the direction of ion flight. Hadamard transform time-of-flightHadamard transform TOF mass spectrometry is a method that can be used with a continuous ion source.[6] Tandem TOF/TOFTOF/TOF is a tandem mass spectrometry method where two time-of-flight mass spectrometers are used consecutively.[7][8][9][10] The first TOF-MS is used to separate the precursor ions, and the second TOF-MS analyzes the product ions. An ion gate for selecting the precursor ion, an ion fragmentation region (e.g. a collision cell) and an ion accelerator may be provided between the first and second TOFMS[11]. DetectorsA time-of-flight mass spectrometer (TOFMS) consists of a mass analyzer and a detector. An ion source (either pulsed or continuous) is used for lab-related TOF experiments, but not needed for TOF analyzers used in space, where the sun or planetary ionospheres provide the ions. The TOF mass analyzer can be a linear flight tube or a reflectron. The ion detector generally consists of microchannel plate detector and a time to digital converter (TDC) or a fast analog-to-digital converter (ADC). Time-to-digital converters register the arrival of a single ion at discrete time "bins"; thresholding discriminates between noise and ion events. The mass spectrum is created by summing a large number of mass spectra. The TDC is an ion counting detector -- it is fast, but dynamic range is limited by the ability of the TDC to react to multiple ion events. Fast analog-to-digital converters measure ion current at discrete time intervals. An ADC detector has a higher dynamic range than the TDC detector, but requires careful impedance matching to minimize detector "ringing". ApplicationsMatrix-assisted laser desorption ionization (MALDI) is a pulsed ionization technique that is readily compatible with TOFMS.See alsoReferences
Bibliography
|
|
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Time-of-flight_mass_spectrometry". A list of authors is available in Wikipedia. |
- Anunciados os vencedores do WITec Paper Award 2025 - Os prémios deste ano recompensam os cientistas pelo seu excelente trabalho na análise de materiais, na investigação de pilhas e na ciência médica
- Annunciati i vincitori del WITec Paper Award 2025 - I premi di quest'anno premiano gli scienziati per il loro eccellente lavoro nell'analisi dei materiali, nella ricerca sulle batterie e nella scienza medica
- Timeline_of_biology_and_organic_chemistry
- Antylia Scientific - Montréal, Canadá
- Scam s.p.a. - Turin, Italie





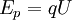 [1]
[1]
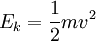 [2]
[2]
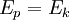 [3]
[3]
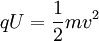 [4]
[4]
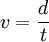 [5]
[5]
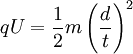 [6]
[6]
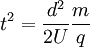 [7]
[7]
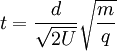 [8]
[8]
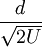 contains constants that in principle do not change when a set of ions are analyzed in a single pulse of acceleration. Eqn 8 can thus be given as:
contains constants that in principle do not change when a set of ions are analyzed in a single pulse of acceleration. Eqn 8 can thus be given as:
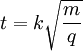 [9]
[9]
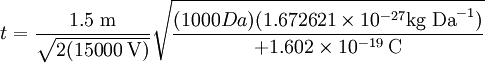 [10]
[10]