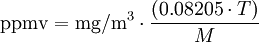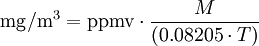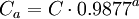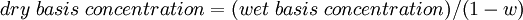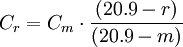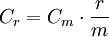To use all functions of this page, please activate cookies in your browser.
my.chemeurope.com
With an accout for my.chemeurope.com you can always see everything at a glance – and you can configure your own website and individual newsletter.
- My watch list
- My saved searches
- My saved topics
- My newsletter
Useful conversions and formulas for air dispersion modelingVarious governmental agencies involved with environmental protection and with occupational safety and health have promulgated regulations limiting the allowable concentrations of gaseous pollutants in the ambient air or in emissions to the ambient air. Such regulations involve a number of different expressions of concentration. Some express the concentrations as ppmv and some express the concentrations as mg/m³, while others require adjusting or correcting the concentrations to reference conditions of moisture content, oxygen content or carbon dioxide content. This article presents a set of useful conversions and formulas for air dispersion modeling of atmospheric pollutants and for complying with the various regulations as to how to express the concentrations obtained by such modeling.[1] Product highlight
Converting air pollutant concentrationsThe conversion equations depend on the temperature at which the conversion is wanted (usually about 20 to 25 degrees Celsius). At an ambient air pressure of 1 atmosphere (101.325 kPa), the general equation is: and for the reverse conversion:
Notes:
Correcting concentrations for altitudeAtmospheric pollutant concentrations expressed as mass per unit volume of atmospheric air (e.g., mg/m³, µg/m³, etc.) at sea level will decrease with increasing altitude because the atmospheric pressure decreases with increasing altitude. The change of atmospheric pressure with altitude can be obtained from this equation:[2]
Given an atmospheric pollutant concentration at an atmospheric pressure of 1 atmosphere (i.e., at sea level altitude), the concentration at other altitudes can be obtained from this equation:
As an example, given a concentration of 260 mg/m³ at sea level, calculate the equivalent concentration at an altitude of 1,800 meters: Ca = 260 × 0.9877 18 = 208 mg/m³ at 1,800 meters altitude Standard conditions for gas volumesA normal cubic meter (Nm³ ) is the metric expression of gas volume at standard conditions and it is usually (but not always) defined as being measured at 0 °C and 1 atmosphere of pressure. A standard cubic foot (scf) is the USA expression of gas volume at standard conditions and it is often (but not always) defined as being measured at 60 °F and 1 atmosphere of pressure. There are other definitions of standard gas conditions used in the USA besides 60 °F and 1 atmosphere. That being understood: 1 Nm³ of any gas (measured at 0 °C and 1 atmosphere of absolute pressure) equals 37.326 scf of that gas (measured at 60 °F and 1 atmosphere of absolute pressure). 1 kmol of any ideal gas equals 22.414 Nm³ of that gas at 0°C and 1 atmosphere of absolute pressure ... and 1 lbmol of any ideal gas equals 379.482 scf of that gas at 60 °F and 1 atmosphere of absolute pressure. Notes:
Windspeed conversion factorsMeteorological data includes windspeeds which may be expressed as statute miles per hour, knots, or meters per second. Here are the conversion factors for those various expressions of windspeed: 1 m/s = 2.237 statute mile/h = 1.944 knots Note:
Correcting for reference conditionsMany environmental protection agencies have issued regulations that limit the concentration of pollutants in gaseous emissions and define the reference conditions applicable to those concentration limits. For example, such a regulation might limit the concentration of NOx to 55 ppmv in a dry combustion exhaust gas corrected to 3 volume percent O2. As another example, a regulation might limit the concentration of particulate matter to 0.1 grain per standard cubic foot (i.e., scf) of dry exhaust gas corrected to 12 volume percent CO2. Environmental agencies in the USA often denote a standard cubic foot of dry gas as "dscf" or as "scfd". Likewise, a standard cubic meter of dry gas is often denoted as "dscm" or "scmd" (again, by environmental agencies in the USA). Correcting to a dry basisIf a gaseous emission sample is analyzed and found to contain water vapor and a pollutant concentration of say 40 ppmv, then 40 ppmv should be designated as the "wet basis" pollutant concentration. The following equation can be used to correct the measured "wet basis" concentration to a "dry basis" concentration:[3]
Thus, a wet basis concentration of 40 ppmv in a gas having 10 volume percent water vapor would have a dry basis concentration = 40 ÷ ( 1 - 0.10 ) = 44.44 ppmv. Correcting to a reference oxygen contentThe following equation can be used to correct a measured pollutant concentration in an emitted gas (containing a measured O2 content) to an equivalent pollutant concentration in an emitted gas containing a specified reference amount of O2:[4]
Thus, a measured NOx concentration of 45 ppmv (dry basis) in a gas having 5 volume % O2 is Correcting to a reference carbon dioxide contentThe following equation can be used to correct a measured pollutant concentration in an emitted gas (containing a measured CO2 content) to an equivalent pollutant concentration in an emitted gas containing a specified reference amount of CO2:[5]
Thus, a measured particulates concentration of 0.1 grain per dscf in a gas that has 8 volume % CO2 is Notes:
See also
References
Categories: Chemical engineering | Environmental engineering |
|||||||||||||||||||||||||||||||||||||||
| This article is licensed under the GNU Free Documentation License. It uses material from the Wikipedia article "Useful_conversions_and_formulas_for_air_dispersion_modeling". A list of authors is available in Wikipedia. |